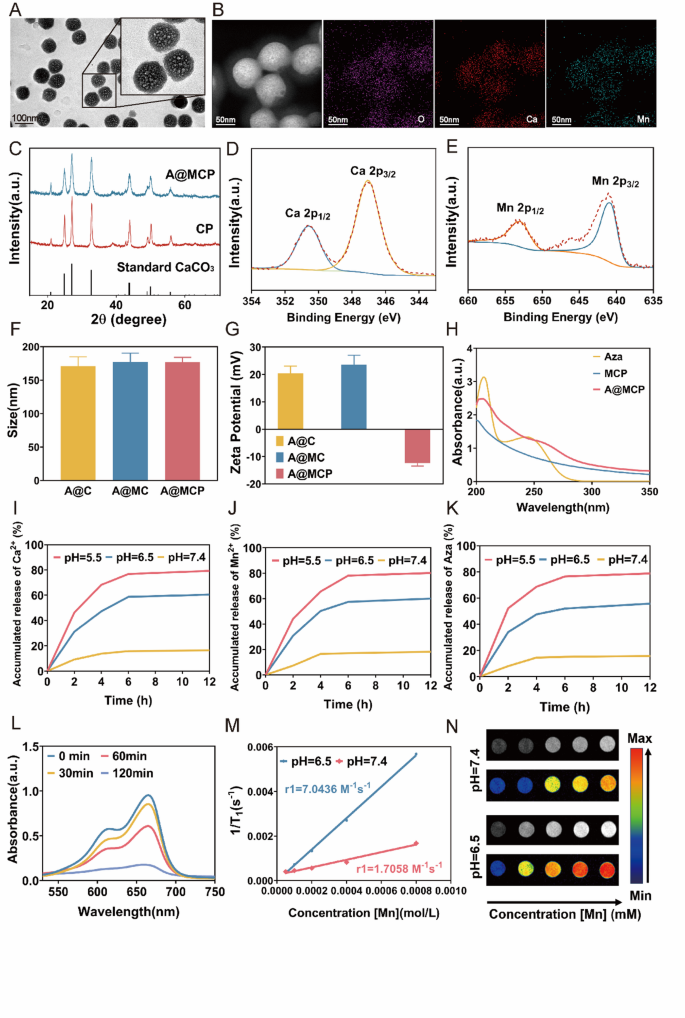
Synthesis and characterization of Aza@Mn-CaCO3-PEG nanoparticles
PEGylated Aza@Mn-CaCO3 nanoparticles have been synthesized utilizing a modified fuel diffusion technique primarily based on earlier research [49, 50]. The NPs exhibited a monodisperse, spherical construction with a uniform measurement of roughly 119.3 ± 8.8 nm, as confirmed by transmission electron microscopy (TEM) (Fig. 1A). Elemental mapping (Fig. 1B) verified the uniform distribution of Ca and Mn on the nanoparticle floor, with atom ratios of 11.4 ± 0.3% and a couple of.6 ± 0.2%, respectively. The X-ray diffraction (XRD) patterns confirmed profitable synthesis of CaCO3. Equally, A@MCP NPs confirmed similar diffraction peaks to CaCO3, indicating that Mn doping and Aza loading had minimal influence on the crystal construction (Fig. 1C). Furthermore, the existence of Ca2+ and Mn2+ parts have been additionally confirmed by X-ray photoelectron spectroscopy (XPS) evaluation (Determine S1) with Mn 2p1/2 and Mn 2p3/2 binding vitality round 653.03 eV and 640.88 eV in addition to the Ca 2p1/2 and Ca 2p3/2 round 350.58 eV and 347.05 eV, respectively (Fig. 1D and E), verifying the profitable doping of Mn2+ into the CaCO3 NPs. After PEGylation, the hydrodynamic measurement of A@MCP NPs, measured by dynamic mild scattering (DLS), was 176.9 ± 5.8 nm, with a polydispersity index of 0.065 ± 0.020 and the zeta potential reversed to roughly − 12.4 mV (Fig. 1F and G and Determine S2). UV-vis absorption spectrum of A@MCP NPs displayed attribute peaks of Aza, confirming profitable encapsulation of Aza (Fig. 1H). The Aza loading content material, calculated from UV-Vis commonplace curves (Determine S3), was roughly 18.6%.
The pH-responsive launch conduct of A@MCP NPs was investigated at completely different pH ranges (Determine I, J, Okay). The decomposition charges of A@MCP accelerated because the pH decreased from 7.4 to five.5 and exhibited time-dependent degradation in acidic options, with cumulative launch charges of Ca2+, Mn2+, and Aza reaching round 76.8%, 78.2%, and 68.7% after 6 h at pH 5.5, respectively. TEM evaluation (Determine S4) additionally revealed the entire collapse of the A@MCP NPs at pH 5.5, indicating the excessive pH-responsive degradation sensitivity of A@MCP NPs.
The power of Mn2+ to generate poisonous hydroxyl radicals (·OH) through a Fenton-like response was accessed utilizing methylene blue (MB) as an indicator. After incubation with A@MCP and H2O2, the absorbance of MB progressively decreased over time, indicating that Mn2+ successfully reacts with H2O2 to generate ·OH, amplifying ROS (Fig. 1L).
The TME-stimulated MRI distinction impact of A@MCP was evaluated by measuring the longitudinal leisure time (T1) in PBS beneath completely different pH. With rising launch of paramagnetic Mn2+, the r1 worth of NPs rose considerably from 1.71 mM− 1 s− 1 at pH 7.4 to 7.04 mM− 1 s− 1 at pH 6.5 (Fig. 2M). Moreover, the T1 MRI distinction sign elevated with increased NP concentrations at pH 6.5 in comparison with pH 7.4 (Fig. 1N), indicating the potential of A@MCP as a pH-activatable T1 MRI distinction agent.
Lastly, in vitro hemolysis assessments have been carried out to evaluate the biocompatibility of A@MCP. After incubation with crimson blood cells for six h, no vital hemolytic exercise was noticed throughout varied concentrations (Determine S5). Even at 200 µg mL− 1, the hemolysis charge remained low at roughly 2.3%, effectively under the allowable restrict of 5%. Moreover, A@MCP was well-dispersed in 10% fetal bovine serum (FBS) for one month (Determine S6). The detachment of PEG was quantified by monitoring modifications of fluorescence indicators in several physiological circumstances at 37 °C [51, 52]. Particularly, DSPE-mPEG-Rhodamine B (DSPE-mPEG-RhB) was used to organize Aza@Mn–CaCO₃–PEG/RB nanoparticles, which have been then incubated at 37 °C with 10% FBS, human serum albumin (HSA, 5 mg/mL), and mouse plasma. At designated time factors, the fluorescence spectra of the samples have been recorded utilizing a microplate reader to observe PEG detachment. As proven in Figures S7–S9, the fluorescence remained secure over 24 h in all examined circumstances, confirming the robustness and retention of PEG on A@MCP nanoparticles beneath physiological circumstances. These outcomes indicated that A@MCP had favorable biocompatibility, providing worthwhile insights for in vivo functions.
Synthesis and characterization of Aza@Mn-CaCO3-PEG nanoparticles. A) TEM photographs of Aza@Mn-CaCO3-PEG NPs. Scale bar, 100 nm; B) Elemental distribution mapping of A@MCP NPs. Scale bar, 50 nm; C) XRD patterns of A@MCP and CP NPs. XPS spectra of D) Ca 2p and E) Mn 2p. F) Hydrated particle measurement modifications of A@C, A@MC and A@MCP (n = 3 for every group). G) Zeta potential modifications of A@C, A@MC and A@MCP (n = 3 for every group). H) The absorption spectra of Aza, MCP and A@MCP. I) Ca2+, J) Mn2+, and Okay) Aza launched from A@MCP at completely different pH. L) UV/vis spectra of MB aqueous answer after degradation by A@MCP for two h. M) T1 leisure charge of A@MCP NPs in several concentrations at completely different pH and N) the corresponding T1-weighted MR photographs. Aza: Azacitidine; CP: CaCO3-PEG; MCP: Mn-CaCO3-PEG; A@MCP: Aza@Mn-CaCO3-PEG; A@C: Aza@CaCO3; A@MC: Aza@Mn-CaCO3
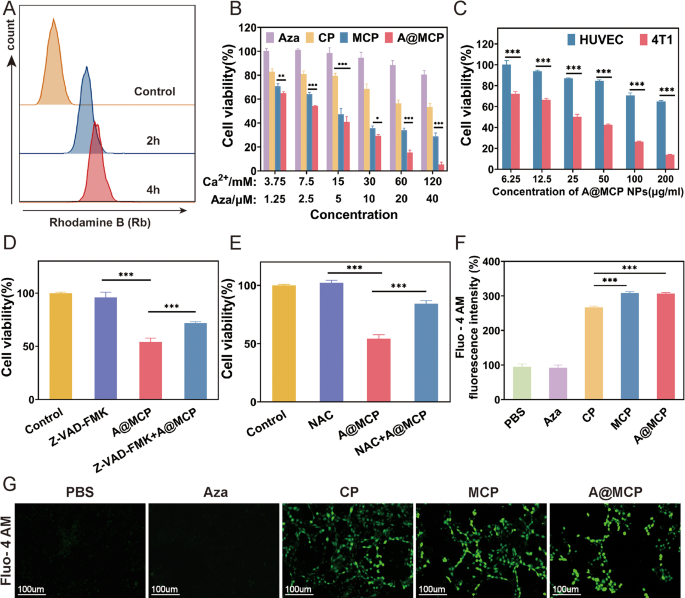
In vitro uptake and cytotoxicity in most cancers cells
Environment friendly uptake of A@MCP NPs is essential for his or her organic exercise. Rhodamine B (Rhb) labeled A@MCP NPs have been utilized to evaluate the mobile uptake utilizing move cytometry and a time-dependent enhance in Rb fluorescence depth was noticed in 4T1 cells inside 4 h (Fig. 2A and Determine S10), indicating profitable supply of the NPs.
Subsequent, cytotoxicity of NPs was evaluated utilizing a cell counting kit-8 (CCK-8) assay (Fig. 2B). Because it has been reported, high-dose Aza all the time features as a chemotherapeutic agent, whereas low doses Aza is utilized for mobile genetics regulation with restricted toxicity [24, 53]. CCK-8 assay outcomes confirmed that Aza exhibited low toxicity, even at a focus of 40 µmol L⁻¹, suggesting that Aza loaded in A@MCP NPs was at a low dose and had no direct cytotoxic results, solely serving primarily for GSDME regulation. In distinction, CP, MCP and A@MCP considerably inhibited the proliferation of 4T1 cells in a concentration-dependent method, with A@MCP exhibiting superior antitumor efficacy. Notably, A@MCP NPs exhibited restricted cytotoxicity in direction of regular cells equivalent to human umbilical vein endothelial cells (HUVECs), sustaining roughly 65% viability whereas 4T1 cell viability considerably dropped to solely about 13% (Fig. 2C). These outcomes indicated the great biocompatibility and particular antitumor exercise of A@MCP NPs as a radiosensitizer.
Moreover, a pan-caspase inhibitor, Z-VAD-FMK and a reactive oxygen species (ROS) scavenger (N-acetyl-L-cysteine, NAC) have been used on 4T1 cells to elucidate the underlying mechanisms of A@MCP-induced cell demise (Fig. 2D and E). Remedy with both Z-VAD-FMK or NAC partially restore mobile exercise, suggesting the involvement of caspase-dependent pathways and ROS in A@MCP-induced cell demise. Moreover, evaluation utilizing Fluo-4 AM probe revealed substantial intracellular Ca²⁺ accumulation in cells handled with CaCO3-based NPs, with Mn-doping selling even better Ca²⁺ inflow and intensifying Ca²⁺ overload (Fig. 2F and G and Determine S11). These findings steered the potential of A@MCP NPs for environment friendly most cancers remedy by synergistic ROS era and Ca²⁺ overload.
In vitro uptake and cytotoxicity in most cancers cells. A) Stream cytometry evaluation of 4T1 cells incubated with A@MCP marked by Rb at completely different time factors. B) Cell viability of 4T1 cells handled completely different therapies for twenty-four h. C) Cell viability of HUVECs and 4T1 cells after incubation with A@MCP NPs for twenty-four h, respectively. Cell viability of 4T1 cells pre-treated with D) Z-VAD-FMK and E) NAC earlier than incubation with A@MCP NPs, respectively (imply ± SD). F) Quantitative move cytometry evaluation (imply ± SD) and G) fluorescence microscopy photographs of Fluo-4AM-stained 4T1cells after varied therapies. Scale bar, 100 μm; Aza: Azacitidine; CP: CaCO3-PEG; MCP: Mn-CaCO3-PEG; A@MCP: Aza@Mn-CaCO3-PEG. The statistical significance was obtained utilizing a one-way ANOVA (n = 3 for every group, *P < 0.05, ** P < 0.01 and *** P < 0.001)
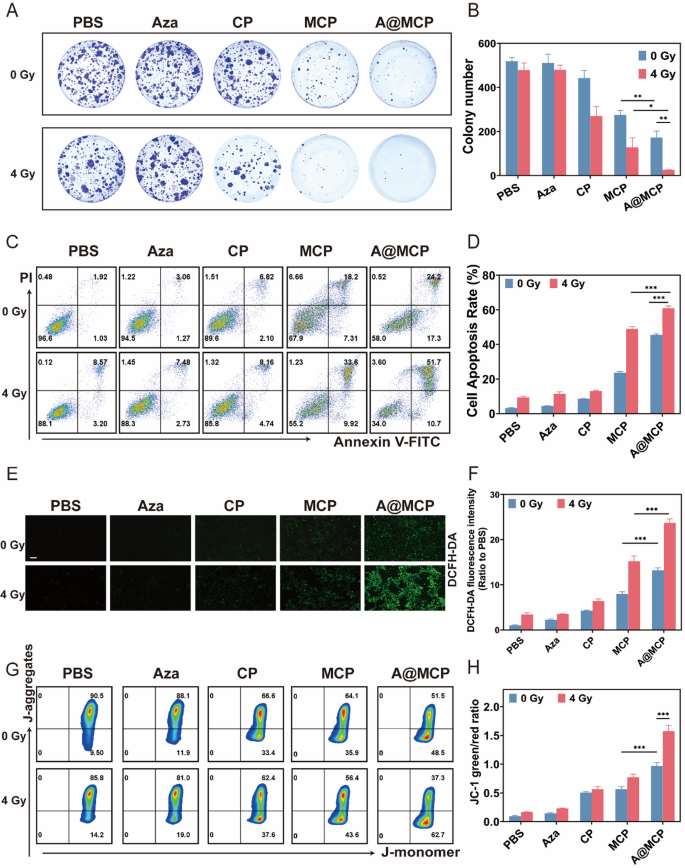
In vitro antitumor actions of A@MCP-sensitized radiotherapy
The radiosensitizing potential of A@MCP NPs was subsequently investigated utilizing a colony formation assay (Fig. 3A and B). With out receiving RT, the cloning effectivity of 4T1 cells handled with A@MCP clearly declined, yielding a survival fraction of roughly 33.2%, which was notably decrease than that in CP (about 85.3%) and MCP (about 52.9%). In distinction, following RT remedy, the survival fraction of CP-treated cells decreased to round 52.0%, whereas MCP additional lowered to round 24.6%. A@MCP remedy confirmed essentially the most pronounced impact, reducing the survival fraction to simply about 5.0%, confirming the substantial radiosensitization functionality of A@MCP.
An apoptosis/necrosis assay was additional carried out to guage A@MCP-mediated radiosensitization in 4T1 cells utilizing move cytometry (Fig. 3C and D). Per the colony formation assay, even with out RT, MCP and A@MCP therapies significantly elevated the extent of cell apoptosis (Annexin V+) to roughly 23.7% and 45.5% respectively. Upon RT publicity, the apoptosis charge within the MCP-treated group rose considerably to roughly 46.7%, whereas the A@MCP + RT group exhibited the markedly highest proportion of apoptotic cells at about 60.9%. These outcomes confirmed the contribution of A@MCP in enhancing the therapeutic efficacy of RT and indicated the synergistic radiosensitization results Ca2+, Mn2+, and Aza.
As indicated beforehand, A@MCP-mediated cell demise might be considerably attenuated by ROS scavenger, suggesting the radiosensitization impact might depend on the oblique destruction of most cancers cells by ROS. Therefore, intracellular ROS era was measured utilizing the DCFH-DA probe through fluorescence microscopy and move cytometry to research the mechanisms underlying the improved radiosensitization by A@MCP (Fig. 3E and F and Determine S12). With out RT, cells handled by MCP NPs confirmed elevated intracellular ROS manufacturing in contrast with the CP group, highlighting the twin ROS amplification results of Ca2+ overload and Mn2+-mediated Fenton-like reactions. Notably, A@MCP remedy resulted in even increased intracellular fluorescence of ROS than the MCP teams, suggesting the inclusion of Aza additional intensified ROS era and exacerbated DNA stress. Upon RT publicity, a pointy enhance in ROS fluorescence was detected in each MCP and A@MCP teams and A@MCP + RT remedy confirmed the very best cumulative ROS ranges amongst all teams. These findings confirmed the synergistic contribution of Ca2+, Mn2 and Aza, together with RT, established a constructive suggestions loop for ROS amplification, additional selling ROS-mediated radiosensitization and cell demise.
The buildup of ROS sometimes accompanies dysfunction of the mitochondrial membrane potential (MMP) [54, 55]. Thus, a JC-1 fluorescent probe was employed to evaluate the results of A@MCP-mediated radiosensitization on mitochondrial operate (Fig. 3G and H and Determine S13). As proven in Fig. 3H, with out RT, the fraction of intracellular aggregates in cells handled with A@MCP NPs was considerably lowered in comparison with CP and MCP teams, indicating the improved mitochondrial dysfunction synergistically resulted by Ca2+, Mn2 and Aza. Notably, after RT at 4 Gy, A@MCP + RT group exhibited the very best fraction of intracellular monomers in cells with the bottom MMP, confirming A@MCP-sensitized radiotherapy induced oxidative stress and causes extreme mitochondrial harm, effectively eliminating most cancers cells.
In vitro antitumor actions of A@MCP-sensitized radiotherapy. A) Consultant photographs of B) colony formation assay of 4T1 cells incubated with PBS, Aza, CP, MCP and A@MCP with or with out RT (4 Gy) (imply ± SD). C) Stream cytometry evaluation of 4T1 cell apoptosis and D) the corresponding apoptosis proportion (imply ± SD). E) Fluorescence microscopy photographs and F) the corresponding quantitative evaluation of 4T1 cells with completely different therapies for six h after which stained with ROS probes (DCFH-DA, inexperienced and imply ± SD). Scale bar, 100 μm; G) Stream cytometry evaluation of mitochondrial membrane potential detected through the use of JC-1 after completely different therapies and H) the corresponding quantitative evaluation (imply ± SD). Aza: Azacitidine; CP: CaCO3-PEG; MCP: Mn-CaCO3-PEG; A@MCP: Aza@Mn-CaCO3-PEG. The statistical significance was obtained utilizing a one-way ANOVA (n = 3 for every group, *P < 0.05, ** P < 0.01 and *** P < 0.001)
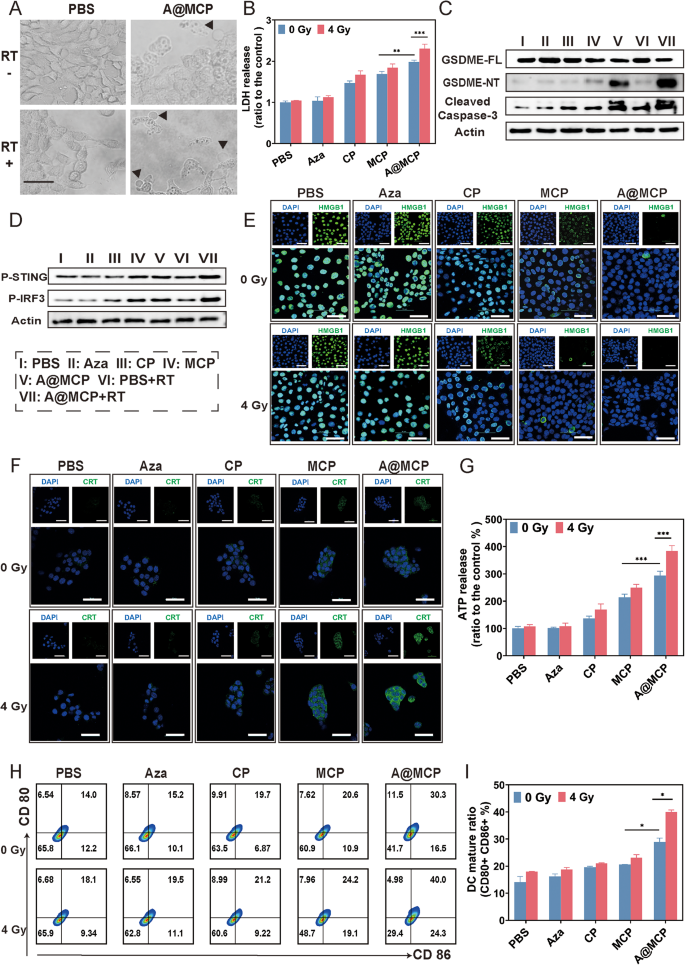
In vitro analysis of pyroptosis and cGAS-STING activation mediated immune activation
Remedy of 4T1 cells with A@MCP, as proven in Fig. 4A utilizing mild microscopy (LM), resulted in mobile swelling adopted by blistering of the cell membrane (arrow), finally resulting in lysis (Fig. 4A). These pyroptotic options grew to become extra pronounced after publicity to RT at 4 Gy, suggesting that A@MCP might function a radiosensitizer, enhancing RT by selling pyroptosis.
Intracellular lactate dehydrogenase (LDH) was launched into the TME on account of cell lysis (Fig. 4B). Align with the LM observations, with out RT publicity, LDH was clearly elevated within the A@MCP remedy and better than that in CP and MCP remedy teams, implying the crucial position of Aza in selling pyroptosis. In the meantime, following RT, cells handled with A@MCP + RT group exhibited considerably highest LDH launch, indicating that A@MCP-enhanced RT-induced cell lysis and demise through pyroptosis.
Western blot evaluation was carried out to discover the mechanism of pyroptosis induced by A@MCP NPs beneath RT (Fig. 4C and Determine S14–S15). Extreme ROS-mediated caspase-3 activation have been recognized as upstream indicators for pyroptosis [13, 16].In the meantime excessive GSDME expression acts as a vital change within the subsequent caspase-3-mediated pyroptotic pathway, resulting in the discharge of its N-terminal fragment (GSDME-N) into the cytoplasm upon activation [13, 14, 22]. Though Aza remedy certainly elevated GSDME-FL expression, it failed to boost GSDME-NT expression and didn’t induce pore formation required for pyroptosis. This commentary steered that GSDME upregulation alone was inadequate to set off pyroptosis within the absence of caspase-3 activation as Aza lacked the capability to induce oxidative stress or mitochondrial harm. In distinction, A@MCP remedy considerably elevated the expression of GSDME-NT and caspase-3 cleavage in comparison with the CP and MCP teams. This discovering underscore the need of ROS–induced caspase-3 activation to attain efficient GSDME cleavage and full pyroptotic execution. Following RT, A@MCP + RT remedy additional upregulated the expression of pyroptosis associated proteins, indicating a pronounced synergistic impact.
To additional dissect the potential of A@MCP/RT induced pyroptosis, a not too long ago recognized extremely particular GSDME inhibitor, methylcobalamin (MeCbl) [56], was noticed to extend cell viability in A@MCP and A@MCP/RT-treated cells, offering preliminary proof the useful relevance of GSDME in A@MCP/RT-induced pyroptosis (Determine S16). After pretreatment with the GSDME-specific inhibitor MeCbl on cells, GSDME cleavage was markedly suppressed in each the A@MCP and A@MCP + RT teams. Notably, the A@MCP + RT group confirmed a extra pronounced suppression of GSDME cleavage in comparison with the A@MCP group, with out affecting cleaved caspase-3 ranges (Determine S17–S19). Collectively, these findings display that A@MCP, comprising Ca²⁺, Mn²⁺, and Aza, synergistically enhances RT-induced activation of the caspase-3–GSDME pyroptotic pathway, providing a potent technique for amplifying cell demise.
Along with inducing pyroptosis, A@MCP mixed with RT enhanced immunoregulatory capability by stimulating the innate immune response via cGAS-STING pathway activation. Western blot evaluation confirmed even with out RT, elevated phosphorylation of STING and IRF3 have been noticed each in MCP and A@MCP therapies, supporting position of Mn2+in enhancing STING pathway activation (Fig. 4D, and Determine S20–S21). Notably, following RT at 4 Gy, the A@MCP + RT group exhibited considerably highest ranges of p-STING and p-IRF3 amongst different teams, indicating the radioimmunity-sensitization capability of A@MCP by synergistically augmenting the STING pathway together with RT.
In the meantime, the relative contribution of Mn²⁺ and RT to cGAS–STING signaling pathway activation was additional in contrast on 4T1 cells (Determine S22–S24). A@MCP remedy considerably upregulated each P-STING and P-IRF3 ranges in comparison with A@CP and PBS teams, indicating that Mn²⁺ is enough to partially activate the STING pathway. RT alone (PBS + RT) produced a modest enhance in STING signaling, possible via radiation-induced cytosolic dsDNA accumulation. Nonetheless, A@MCP + RT remedy resulted within the highest expression ranges of each P-STING and P-IRF3, exceeding all different teams. These findings re-emphasized cooperative roles between Mn²⁺ and RT in STING pathway enhancement.
Radiotherapy has been proven to set off ICD by exposing tumor antigens and releasing danger-associated molecular patterns (DAMPs), thus stimulating anti-tumor immune response [57, 58]. A@MCP-mediated radiosensitization, pushed by pyroptotic cell demise, might additional improve RT-induced ICD for improved radioimmunotherapy outcomes. The publicity of calreticulin (CRT) and the discharge of excessive mobility group field 1 (HMGB1) and ATP have been analyzed as indicators of ICD to verify this enhancement (Fig. 4E, 4F and 4G, and Determine S25–S26). Thereinto, with out RT, cells handled with A@MCP confirmed extra apparent HMGB1 translocation, CRT publicity and ATP launch than CP and MCP remedy, indicating the mixed enhanced pyroptotic results of Ca²⁺, Mn²⁺, and Aza led to extra pronounced ICD outcomes. After RT, essentially the most minimal nuclear fluorescence of HMGB1, essentially the most intense fluorescence on the cell floor of CRT and the very best launch of ATP have been detected in cells handled within the A@MCP + RT group, additional implying the synergistic impact of A@MCP in enhancing immunogenicity and selling immune-stimulatory cell demise via the mix of pyroptosis and RT.
Enhanced cGAS-STING pathway activation and ICD induction may promote antigen presentation and dendritic cells (DC) maturation. Stream cytometry evaluation of CD80 and CD86 markers revealed a major enhance of mature DCs within the A@MCP group in contrast with CP and MCP remedy, indicating that the synergistic impact of the mix of pyroptosis and STING activation to advertise DC maturation. After RT at 4 Gy, the A@MCP remedy exhibited the very best mature DC proportion, reaching roughly 39.9%, considerably increased than CP + RT and MCP + RT teams (Fig. 4H and I), suggests the potential radioimmunity-sensitivity of A@MCP to drive DC maturation and provoke efficient immune activation.
In abstract, these outcomes affirm that A@MCP NP sensitized RT by pyroptosis and cGAS-STING pathway to evoke sturdy immune responses and enhance the efficacy of radioimmunotherapy.
Pyroptosis and cGAS-STING activation co-mediated immune activation. A) Consultant vibrant subject microscopy picture of 4T1 cells after remedy. The arrows point out pyroptotic cells; Scale bar, 100 μm; B) LDH launch ranges of 4T1 cells following completely different therapies (imply ± SD) C) The expression of the pyroptosis-related proteins in 4T1 cells following completely different therapies measured by western blot assay. D) The expression of the STING-related proteins in 4T1 cells following completely different therapies measured by western blot assay. E) CLSM photographs of HMGB1 expression (scale bars: 50 μm). F) CLSM photographs of CRT expression (scale bars: 50 μm). G) ATP launch ranges of 4T1 cells following completely different therapies (imply ± SD). H) Stream cytometry evaluation of DC maturation after completely different therapies and I) the corresponding quantitative evaluation (imply ± SD). Aza: Azacitidine; CP: CaCO3-PEG; MCP: Mn-CaCO3-PEG; A@MCP: Aza@Mn-CaCO3-PEG. The statistical significance was obtained utilizing a one-way ANOVA (n = 3 for every group, *P < 0.05, ** P < 0.01 and *** P < 0.001)
In vivo MRI imaging and biodistribution profiling of A@MCP for antitumor conduct
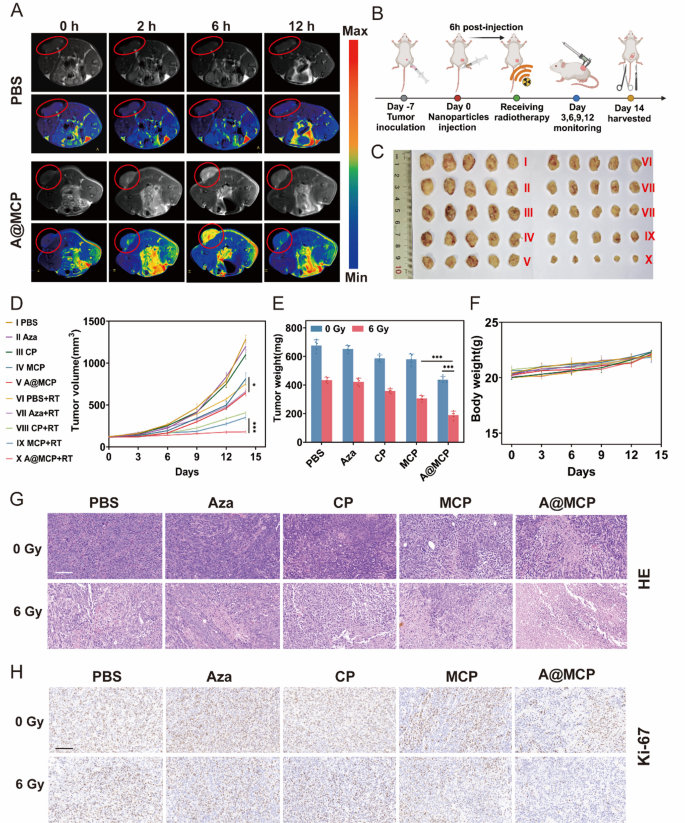
Leveraging its activatable T1 MRI properties, A@MCP was administered to 4T1 tumor-bearing mice to evaluate its tumor-specific imaging potential. As proven in Fig. 5A, no vital T1 sign enhancement was detected within the PBS group. In distinction, the A@MCP group demonstrated a considerably enhanced T1 MRI sign on the tumor web site, peaking 6 h post-administration, with ~ 3.5-fold enhance in comparison with the PBS group (Determine S27). This superior imaging functionality enabled visualization of nanoparticle biodistribution, allowed exact image-guidance for RT window, demonstrating its theragnostic potential for most cancers remedy.
Later, DiR-labeled A@MCP was administrated through tail vein injection, adopted by real-time fluorescence imaging at varied time factors to discover in vivo pharmacokinetic and biodistribution behaviors (Determine S28–S29). Per in vivo MRI imaging, A@MCP sustainably accrued on the tumor web site, reaching peak fluorescence depth at 6 h post-injection, indicating environment friendly and time-dependent tumor enrichment. To quantify the ion retention within the tumor and wholesome organs extra exactly, we additionally measured the quantity of Ca and Mn in tumor-bearing mice utilizing ICP-MS at predetermined time factors as much as 24 h after injection (Determine S30–S31). Per the fluorescence outcomes, A@MCP effectively accrued within the tumor with peak effectivity noticed at 6 h post-injection. In the meantime each Ca²⁺ and Mn²⁺ accrued primarily within the tumor, considerably increased than in different wholesome organs such because the liver, spleen, and kidneys. These outcomes collectively assist the in vivo security of A@MCP and reinforce its potential for tumor-targeted supply with minimal systemic toxicity.
To evaluate the antitumor impact of A@MCP-sensitized radiotherapy, 4T1 tumor-bearing mice have been handled in line with the established protocol (Fig. 5B). Gross observations of the dissected tumors aligned with modifications in tumor quantity (Fig. 5C and D). With out RT, Aza-treated group had no impact on tumor inhibition, whereas the A@MCP-treated group confirmed considerably increased inhibition of tumor progress in contrast with each the CP and MCP teams, suggesting the mixed impact of Aza, Ca2+ and Mn2+ in enhancing tumor suppression. Following RT at 6 Gy, the A@MCP group confirmed essentially the most potent antitumor impact, with an ~ 86.1% discount in tumor progress, a lot increased than that noticed within the CP and MCP teams. Tumor weight additionally confirmed the improved suppression impact of A@MCP mixed with X-ray remedy. These outcomes underscored the antitumor results of A@MCP NPs-sensitized RT and improved the general therapeutic efficacy (Fig. 5E and Determine S32). Moreover, a slight enhance in physique weight throughout all remedy teams signifies the great biosafety of A@MCP (Fig. 5F and Determine S33). H&E and Ki-67 staining additional confirmed that with out RT, extra apparent mobile harm and a better discount in Ki-67 expression have been noticed within the A@MCP remedy group in comparison with the opposite teams. After RT, tumors handled with A@MCP + RT exhibited essentially the most extreme mobile harm and the strongest inhibition of tumor cell proliferation (Fig. 5G and H). These outcomes highlighted the success of mixing Aza, Ca2+ and Mn2+ to sensitize RT in enhancing tumor progress suppression.
In Vivo MRI Imaging guided Antitumor Conduct. A) T1-weighted MRI photographs of 4T1 tumor-bearing mice earlier than and after therapies (n = 3 for every group) and B) Schematic of the in vivo experimental course of for 4T1 tumor-bearing mice. C) Digital photographs of dissected tumors from the mice at day 14 (n = 5 for every group). D) Tumor progress curves of the mice with completely different therapies (imply ± SD, n = 5). E) Dissected tumor weights of the mice with completely different therapies (imply ± SD, n = 5). F) Physique weight variation of the mice through the remedy (imply ± SD, n = 5). G) H&E and H) Ki-67 staining photographs of tumor tissues excised from the mice at day 14. Scale bar: 100 μm; Aza: Azacitidine; CP: CaCO3-PEG; MCP: Mn-CaCO3-PEG; A@MCP: Aza@Mn-CaCO3-PEG. The statistical significance was obtained utilizing a one-way ANOVA (*P < 0.05, ** P < 0.01 and *** P < 0.001)
In vivo analysis of pyroptosis and cGAS-STING Co-activaed immune responses to sensitize RT
To research in vivo immunological response activation, western blot evaluation was carried out to evaluate GSDME-mediated pyroptosis and cGAS-STING pathway activation in tumor tissues. Per in vitro findings, A@MCP considerably upregulated pyroptosis-related proteins, together with cleaved caspase-3 and GSDME-NT, selling extra pyroptosis than free Aza, CP and MCP. After RT at 6 Gy, the A@MCP group confirmed highest stage of cleaved caspase-3 and GSDME-NT (Determine S34–S36), in addition to phosphorylated STING and IRF3 (Determine S37–S39).
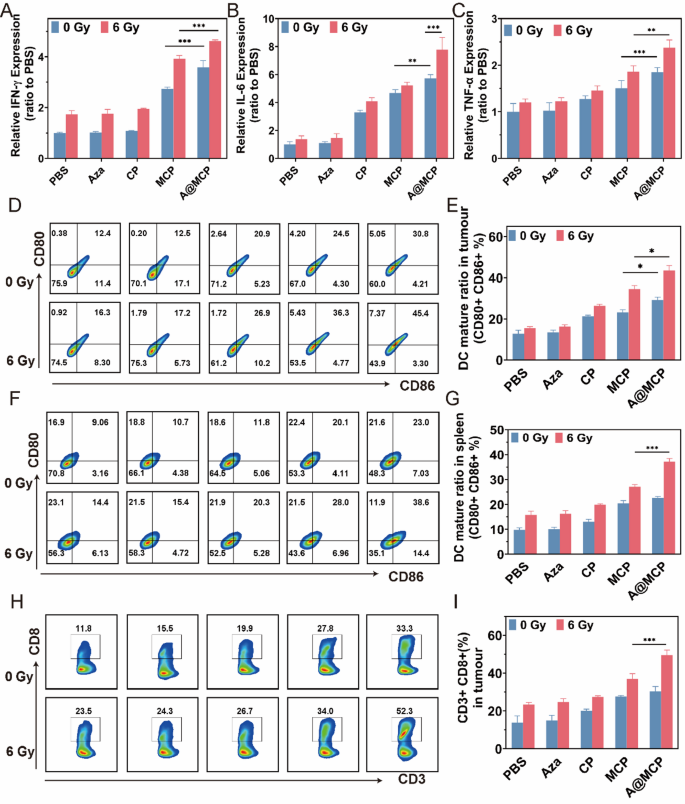
Pyroptosis and STING pathway co-activation are sometimes accompanied by the secretion of pro-inflammatory components, driving immunological responses. Enzyme-linked immunosorbent assay (ELISA) was used to measure IFN-γ, IL-6, and TNF-α ranges in blood supernatants (Fig. 7A, B and C). The A@MCP group confirmed considerably increased ranges of IFN-γ, IL-6, and TNF-α than CP and MCP teams, highlighting the interplay of Aza, Mn²⁺, and Ca²⁺ in enhancing immune activation. Combining RT at 6 Gy, the inflammatory cytokines have been additional upregulated with the A@MCP + RT remedy exhibiting the very best ranges of IFN-γ, IL-6, and TNF-α, exhibiting the capability of A@MCP-sensitized RT to accentuate pro-inflammatory cytokine manufacturing and rework TME from immunosuppressive into immune-stimulating.
To additional distinguish pyroptosis from different cell demise modalities, pyroptosis-specific cytokines IL-1β and IL-18 was additional quantified. As proven in Determine S40–S41, A@MCP remedy considerably elevated each IL-1β and IL-18 ranges in comparison with CP and MCP teams beneath non-irradiated circumstances. Importantly, RT (6 Gy) additional enhanced IL-1β and IL-18 expression, with the A@MCP + RT group exhibiting essentially the most pronounced upregulation, confirming collaborative enhanced pyroptosis-associated inflammatory signaling, in keeping with the noticed activation of the caspase-3–GSDME axis.
Above elevated inflammatory components additional facilitated the activation of DCs after which primed the infiltration of cytotoxic T lymphocytes (CTLs). Three days after RT, tumor and spleen tissues have been harvested to evaluate mature DC and CTL infiltration by move cytometry. Per in vitro outcomes, A@MCP + RT remedy considerably promoted DC maturation in each tumor and spleen with the very best proportion of mature DCs in contrast with different remedy teams (Fig. 6D, E, F and G). Equally, with out RT, elevated proliferation of CD8 + in tumor have been noticed in A@MCP remedy relative to different remedy teams. Following RT, cell handled with A@MCP + RT confirmed the very best A@MCP proportion of CD8 + T cell activation amongst all teams (Fig. 6H and I). In abstract, these in vivo outcomes offered compelling proof that A@MCP as a potent radiosensitizer, leveraging pyroptosis and STING co-activation to sensitize RT to spice up systemic antitumor immune activations and produce improved antitumor responses.
In vivo immunological response activation. The extent of A) IFN-𝛾, B) IL-6, and C) TNF-𝛼 in plasma (imply ± SD) following the indicated therapies on day 14. D) Stream cytometry evaluation of DC maturation from tumors of handled mice and E) the corresponding quantitative evaluation (imply ± SD). F) Stream cytometry evaluation of DC maturation from spleens of handled mice and G) the corresponding quantitative evaluation (imply ± SD). H) Stream cytometry evaluation of intratumor infiltration of CD8 + T cells from tumors of handled mice and I) the corresponding quantitative evaluation (imply ± SD). Aza: Azacitidine; CP: CaCO3-PEG; MCP: Mn-CaCO3-PEG; A@MCP: Aza@Mn-CaCO3-PEG. The statistical significance was obtained utilizing a one-way ANOVA (n = 3 for every group, *P < 0.05, ** P < 0.01 and *** P < 0.001)
Biosafety evaluation of radiosensitizer A@MCP-enhanced radiotherapy
H&E-stained photographs of main organs (coronary heart, liver, spleen, lung, and kidney) from all remedy teams revealed no pathological harm (Determine S42). Moreover, routine blood assessments, together with serum biochemistry markers equivalent to creatine kinase (CK-MB), creatinine (CREA), and alanine aminotransferase (ALT), remained inside regular physiological ranges (Determine S43). These findings display the superb biocompatibility of A@MCP, with no detectable poisonous unintended effects in mice, highlighting its sturdy potential for scientific software.