CXCL12 mediates adipose tissue regeneration by recruiting ASCs through migrasomes
To elucidate the mechanism underlying comfortable tissue regeneration, we utilized our beforehand established fats grafting mannequin with a donor website [23, 24] (Fig. 1A). This mannequin optimizes the exploration on adipose regeneration by higher mimicking the scenario of scientific autologous fats grafting which comprises fats harvesting (donor website) and fats grafting (recipient website). Adipose tissues on the donor and recipient websites on this mannequin confirmed completely different restore and regeneration outcomes, with tissues of the donor website repaired higher than that on the recipient website. And the mechanism behind this discrepant regeneration course of has been proved to be ascribed to the completely different expression of CXCL12 put up tissue harm [24].
Herein, in similar mice mannequin, hematoxylin and eosin (HE) evaluation at 90 days after grafting confirmed that mature adipocytes have been commonly organized, small adipocytes (white arrows) have been scattered all through, and the tissue construction was steady on the donor website, whereas damaged cells, vacuoles (asterisk), and extreme tissue fibrosis have been noticed on the recipient website (Fig. 1B). Western blot evaluation confirmed that protein expression of CXCL12 and CXCR4 was larger on the donor website than on the recipient website as early as Day3 after tissue harm (Fig. 1C-E). Quantitative evaluation of immunofluorescence staining of CD34 confirmed that the variety of CD34+ ASCs on the donor website was larger than that on the recipient website quickly after harm, elevated from Day3 to Day 14, and decreased thereafter. Nonetheless, infiltration of ASCs was delayed on the recipient website. The variety of CD34+ ASCs on the recipient website was decrease than that on the donor website throughout the first 14 days, elevated from Day 14 to Day 30, and decreased thereafter (Fig. 1F). Considerably extra CD34+ ASCs infiltrated the recipient website than the donor website on the later stage of tissue restore, indicating that regeneration was incomplete. Against this, there have been fewer ASCs on the donor website and perilipin staining confirmed that the tissue construction was built-in (Fig. 1G).
To analyze whether or not migrasomes take part in tissue restore, expression of the migrasome markers TSPAN4 and TSPAN7 was examined in tissue samples from Day 3. Immunofluorescence staining confirmed that vesicles enriched with TSPAN4 and TSPAN7 have been current at each the donor and recipient websites. Magnified photos confirmed that a few of these vesicles have been connected to tubular buildings that prolonged from cells, whereas others have been scattered amongst cells (Fig. 1H). The diameters of those vesicles have been largely 0.5–3 μm, which is in line with the characterizations of migrasomes [19,20,21] (Fig. 1I). Moreover, there have been considerably extra migrasome-like vesicles on the donor website than on the recipient website quickly after fats grafting (Fig. 1J).
These findings confirmed that the chemokine CXCL12 would possibly mediate infiltration of ASCs throughout adipose tissue regeneration and that migrasomes is perhaps concerned in expression of CXCL12.
Determine 1. Recruitment of ASCs by CXCL12 mediates adipose tissue regeneration. (A) Schematic illustration of the novel mouse fats grafting mannequin with a donor website. N = 7 in every teams. (B) HE staining of adipose tissue obtained from the donor and recipient websites 90 days after surgical procedure. Scale bar, 50 μm. White arrows point out small adipocytes and asterisks point out vacuoles. (C) Western blot evaluation of CXCL12 and CXCR4 protein expression in adipose tissue from the donor and recipient websites 3 days after surgical procedure. (D-E) Semi-quantitative evaluation of CXCL12 and CXCR4 protein expression. *p < 0.05 in contrast with Donor. (F) Quantitative evaluation of infiltrated CD34+ ASCs per area on the donor and recipient website over time. *p < 0.05 in contrast with Donor. (G) Immunofluorescence staining of CD34 and perilipin in adipose tissue from the donor and recipient website at 60 days after surgical procedure. Scale bar, 20 μm. (H) Immunofluorescence stanning of TSPAN4 and TSPAN7 in adipose tissue from donor and recipient websites at 3 days after surgical procedure. Scale bar, 10 μm. (I) Diameters of the TSPAN4+ and TSPAN7+ vesicles. n = 50 vesicles per group. (J) Quantitative evaluation of TSPAN4+ and TSPAN7+ vesicles per area in adipose tissue from the donor and recipient website at 3 days after surgical procedure. *p < 0.05 in contrast with Donor. The information imply ± SEM. Statistical variations in (D), (E), (I) and (J) have been assessed with Pupil’s t take a look at, (F) was analyzed with One-way ANOVA.
CXCL12 regulates recruitment of ASCs throughout uneven adipose tissue regeneration
To additional look into the mechanism behind the completely different expression of CXCL12 between the donor and recipient website, we sought to decrease the bias introduced by physiological setting. Thus, we transferred the adipose regeneration mannequin on the bilateral inguinal fats pads of mice. To additional mimic the harm sample of donor and recipient website, we manipulated the inguinal vessels contemplating essentially the most distinguished distinction between the donor and the recipient website of fats grafting is that the vascular honeycomb construction is unbroken at aspirated websites after liposuction whereas grafts at recipient websites are avascular [26]. This uneven adipose regeneration mannequin was well-established by the printed work to judge the regeneration of adipose tissue and the exercise of ASCs put up tissue harm [27, 28]. To this finish, the inguinal vessels have been left intact on the correct aspect to imitate the donor website (Regular) and reduce with scissors on the left aspect to imitate the recipient website (Ischemic) (Fig. 2A).
As anticipated, macroscopic statement on Day30 confirmed that tissues had a pure look and comfortable texture within the Regular group, however appeared atrophic and brittle within the Ischemic group (Fig. 2B). Histological evaluation confirmed that otherwise sized adipocytes have been tightly organized within the Regular group, whereas giant vacuoles and extreme fibrosis have been noticed within the Ischemic group (Fig. 2C). Immunofluorescence staining of CD34 confirmed that considerably extra ASCs infiltrated tissues within the Regular group than within the Ischemic group as early as Day3 put up harm, and the variety of ASCs within the Regular group stored rising from Day3 to Day7 and decreased thereafter. Nonetheless, infiltration of ASCs was delayed within the Ischemic group. The variety of infiltrated ASCs within the Ischemic group remained comparatively low throughout the first 14 days post-injury and elevated sharply from Day 14 to Day 30 (Fig. 2D, E). Perilipin is barely detectable in viable adipocytes. Staining of perilipin confirmed that the adipose tissue construction was comparatively steady throughout the first 7 days post-injury within the Regular group, whereas the perilipin+ space sharply decreased within the Ischemic group. From Day 7 to Day 14, the perilipin+ space decreased within the Regular group however was stably small within the Ischemic group. From Day 14, tissue regeneration was noticed in each teams, as proven by the elevated perilipin+ space. Nonetheless, Nonetheless, the perilipin+ space was considerably bigger, and the variety of infiltrated ASCs was decrease within the Regular group than within the Ischemic group on Day 30, suggesting that regeneration was higher within the Regular group (Fig. 2D, F).
To additional examine the mechanism underlying the uneven regeneration between the Regular and Ischemic teams, we examined expression of CXCL12 and CXCR4. Quantitative PCR (qPCR) evaluation confirmed that expression of CXCL12 and CXCR4 was larger within the Regular group than within the Ischemic group quickly after harm. Expression of CXCL12 and CXCR4 within the Ischemic group started to extend on Day 7 and surpassed that within the Regular group after Day 14 (Further file 1: Determine S1). Western blot evaluation confirmed that expression of CXCL12 and CXCR4 was considerably larger within the Regular group than within the Ischemic group on Day 7, at which level infiltration of ASCs was evident (Fig. 2G-I). These information present proof that CXCL12 induced early infiltration of ASCs through CXCR4 which mediated the uneven regeneration of adipose tissue.
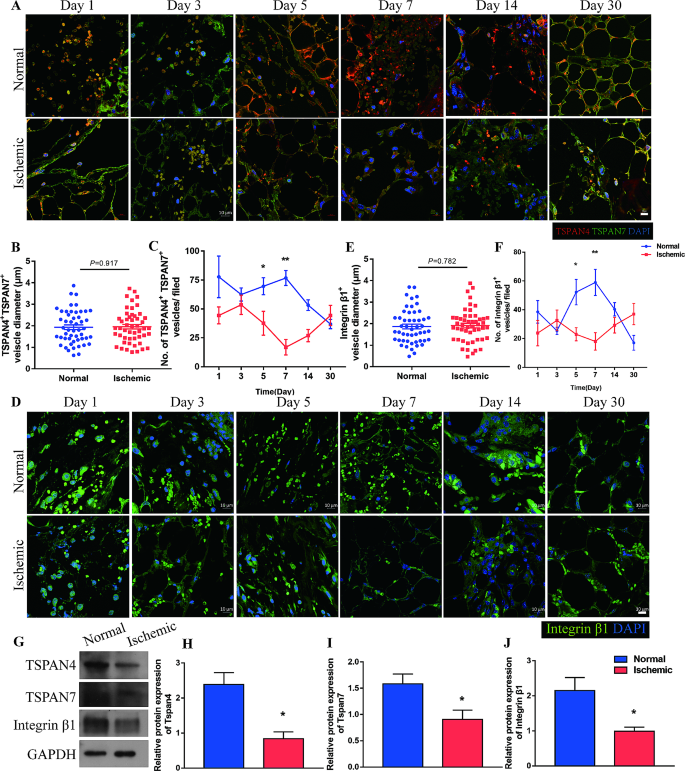
Migrasomes are detected throughout uneven adipose tissue regeneration
Migrating cells combination tetraspanin proteins, particularly TSPAN4, to type a bulbous membrane on the swelling domains of retraction fibers and eventually type migrasomes [29]. Migrasomes can combine spatial and biochemical data, by which cells are recruited to particular location to exert the perform of organ growth or tissue regeneration [19,20,21]. Thus, if migrasomes certainly mediated the institution of CXCL12 sign in adipose tissue regeneration, the variety of migrasomes ought to correlated with the expression of CXCL12 and recruited ASCs in addition to regeneration outcomes between the 2 sides of adipose restore mannequin. To confirm if migrasomes play a task in adipose tissue regeneration, the presence of TSPAN4+ and TSPAN7+ vesicles was examined within the uneven regeneration mannequin. Immunofluorescence staining of tissues detected vesicles enriched with TSPAN4 and TSPAN7 within the extracellular areas or connected to fibers that projected from cells (Fig. 3A). The typical diameter of those vesicles was ∼ 2 μm, which is in line with the diameter of migrasomes (Fig. 3B). The variety of migrasome-like vesicles within the Regular group was larger than that within the Ischemic group as early as Day 1 post-injury, decreased from Day 1 to Day 3, sharply elevated till Day 7, and decreased thereafter. Nonetheless, modifications within the variety of migrasome-like vesicles have been delayed within the Ischemic group. The variety of migrasome-like vesicles within the Ischemic group was decreased at Day 5, elevated from Day 7, and exceeded that within the Regular group after Day 14 (Fig. 3C).
Migrasomes are detected throughout adipose tissue regeneration. (A) Immunofluorescence staining of TSPAN4 and TSPAN7 in adipose tissue from the Regular and Ischemic teams over time. Scale bar, 10 μm. (B) Diameters of the TSPAN4+ and TSPAN7+ vesicles. n = 50 vesicles per group. (C) Quantitative evaluation of TSPAN4+ and TSPAN7+ vesicles per area in adipose tissue from the Regular and Ischemic group over time factors. *p < 0.05, **p < 0.01 in contrast with Regular. (D) Immunofluorescent staining of integrin β1 in adipose tissue from Regular and Ischemic teams over time. Scale bar, 10 μm. (E) Diameters of integrin β1+ vesicles. n = 50 vesicles per group. (F) Quantitative evaluation of integrin β1+ vesicles per area in adipose tissue from the Regular and Ischemic teams over time. *p < 0.05, **p < 0.01 in contrast with Regular. (G) Western blot evaluation of TSPAN4, TSPAN7 and integrin β1 protein expression in adipose tissue from Regular and Ischemic teams at 7 days after surgical procedure. (H-J) Semiquantitative evaluation of TSPAN4, TSPAN7, and integrin β1 protein expression. *p < 0.05 in contrast with Regular. The information are imply ± SEM. Statistical variations in (B), (E), (H), (I) and (J) have been analyzed utilizing Pupil’s t take a look at. (C) and (F) have been assessed by One-way ANOVA
Enrichment of integrin on the underside of migrasomes allows their tethering to the extracellular matrix in order that they don’t transfer away with cell migration and set up a localized sign alongside the migrating pathway of cells [30]. To additional determine the migrasome-like vesicles, integrin β1 was stained. Vesicles enriched with integrin β1 have been noticed within the extracellular house, and a few of these vesicles have been connected to cell projections (Fig. 3D). Integrin β1-enriched vesicles had virtually an identical diameter distributions as TSPAN4+ and TSPAN7+ vesicles, and shared an identical exhibition sample (Fig. 3E, F).
We additional examined expression of migrasome markers in tissues. qPCR evaluation confirmed that expression of TSPAN4, TSPAN7, and integrin β1 was elevated within the Regular group throughout the first 7 days put up harm, however started to extend from Day 7 within the Ischemic group and was larger within the Ischemic group than within the Regular group after Day 14 (Further file 2: Determine S2). Western blot evaluation confirmed that larger protein expression of TSPAN4, TSPAN7, and integrin β1 was considerably larger within the Regular group than within the Ischemic group on Day 7 (Fig. 3G-J).
Therefore, we detected the presence of migrasomes throughout adipose tissue regeneration. As anticipated, the sample of migrasomes resembled that of CXCL12 expression in adipose tissue after harm.
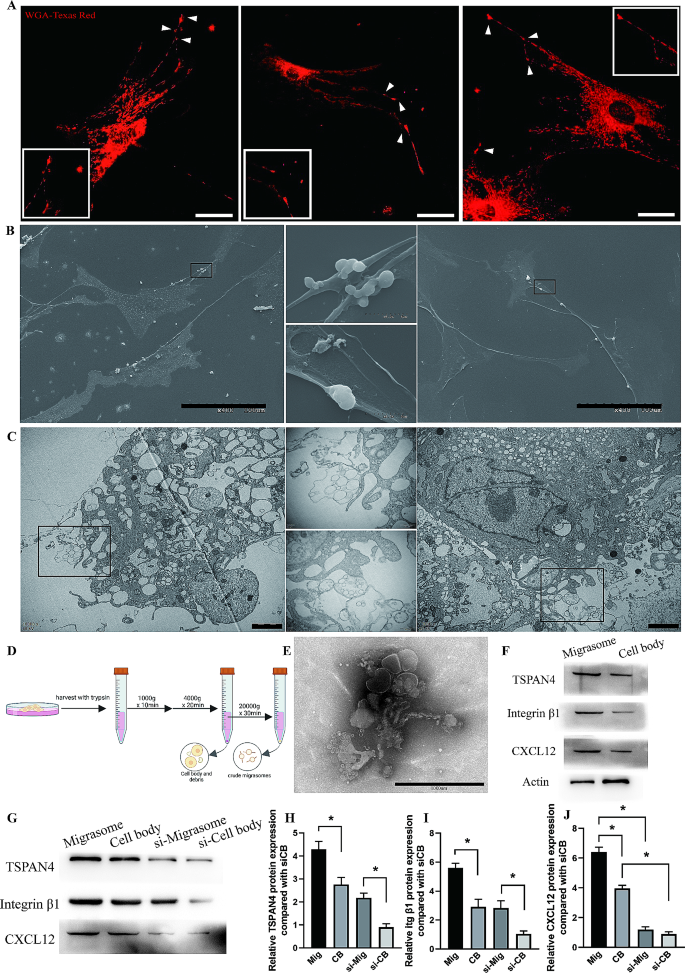
ASCs generate migrasomes
We seen that the sample of migrasomes resembled that of ASCs within the Regular and Ischemic teams (Fig. 2), with infiltration of migrasomes and ASCs delayed within the Ischemic group in contrast the Regular group. ASCs have been documented to secret CXCL12 beneath inflammatory circumstances [31]. We thus postulated that ASCs generate migrasomes. Fluorescently tagged wheat-germ agglutinin (WGA) is a probe for speedy detection of migrasomes in residing cells [32]. We first noticed cultured ASCs stained with WGA-Texas Purple. Confocal microscopy confirmed that quite a few vesicles enriched with WGA with a diameter of ∼ 2 μm have been scattered round ASCs or connected to projections that prolonged from these cells (Fig. 4A). These vesicles have been oval formed, with diameter that matches the definition of migarsomes and clustered on one aspect of the cells, proving these vesicles are migrasomes. Dwell cell time-lapse confocal microscopy additionally revealed the expansion of migrasomes on the ideas of or alongside the retraction fibers generated by ASCs (Further file 3: Determine S3; Supplementary Video 1). Scanning electron microscopy confirmed that ASCs generated membrane-bound vesicles with diameters of 0.5–3 μm that have been connected to the ideas or intersections of retraction fibers, a typical attribute of migrasomes (Fig. 4B). Transmission electron microscopy (TEM) confirmed that the vesicles have been related or in shut proximity to retraction fibers of ASCs. Most of those vesicles have been single-membraned, oval, with the diameter of 0.5–3 μm and contained quite a few small vesicle (Fig. 4C), which is agreed with the definition of migrasomes. Thus, we confirmed that ASCs can generate migrasomes.
ASCs generate migrasomes. (A) Confocal microscopy photos of cultured ASCs stained with WGA-Texas Purple. Scale bar, 10 μm. White arrows indicated vesicles enriched with WGA which are extremely resembled migrasomes, with diameter of ∼ 2 μm and have been scattered round ASCs or connected to projections that prolonged from these cells. (B) Scanning electron microscopy photos of cultured ASCs. Scale bar, 100 μm. (C) TEM photos of ASCs cluster. Scale bar, 2 μm. (D) Schematic illustration of the centrifugation procedures used to isolate migrasomes. (E) TEM picture of remoted migrasomes. Scale bar, 1000 nm. (F) Western blot evaluation of TSPAN4, integrin β1, actin and CXCL12 protein expression in remoted migrasomes. (G) Western blot evaluation of TSPAN4, integrin β1 and CXCL12 protein expression in remoted migrasomes from ASCs or ASCs handled with siRNA. (H-J) Semiquantitative evaluation of TSPAN4, Integrin β1, and CXCL12 protein expression. *p < 0.05. The information are imply ± SEM. Statistical variations have been analyzed utilizing One-way ANOVA adopted by Bonferroni posttest
To substantiate the position of ASC-derived migrasomes in adipose tissue regeneration, we first remoted migrasomes from cultured ASCs (Fig. 4D). The remoted migrasomes have been analyzed by TEM, which confirmed a attribute morphological characteristic an identical to these beforehand reported, with spherical form, attachment to retraction fibers and containing luminal vesicles [19,20,21, 25, 33] (Fig. 4E). Furthermore, the preparations have been extremely enriched with the migrasome markers TSPAN4 and integrin β1, whereas expressed decrease ranges of actin in contrast with cell physique (Fig. 4E). Research have documented the enrichment of CXCL12 inside migrasomes [19,20,21]. Persistently, migrasomes remoted from ASCs contained considerably larger ranges of CXCL12 than the cell our bodies (Fig. 4F). In abstract, we confirmed that ASCs can generate migrasomes enriched with CXCL12.
To confirmed the enrichment of CXCL12 inside migrasomes and the elevated CXCL12 protein expression of migrasomes was certainly mediated by the transferring of CXCL12 from ASCs, we should deplete the endogenous CXCL12 expression of ASCs. Utilizing siRNA considerably decreased the protein expression of CXCL12 in ASCs (Further file 4: Determine S4). Remoted migrasomes from each ASCs and siRNA handled ASCs expressed larger stage of migrasome marker TSPAN4 and Integrin β1 in contrast with cell physique (Fig. 4G-I). Migrasomes contained considerably larger stage of CXCL12 in contrast with cell physique whereas depleting the expression of CXCL12 inside ASCs considerably decreased the CXCL12 stage inside migrasomes, substantiating the direct relationship between migrasomes and CXCL12 in addition to between migrasomes and ASCs (Fig. 4G-J). Moreover, immunofluorescent stained ASCs with WGA and CXCL12 confirmed the colocalized enrichment of CXCL12 with WGA, which straight revealed the supply of CXCL12 by adipose-derived stem cells through migrasomes (Further file 5: Determine S5).
Migrasomes promote ASC recruitment and tissue regeneration through CXCR4/RhoA
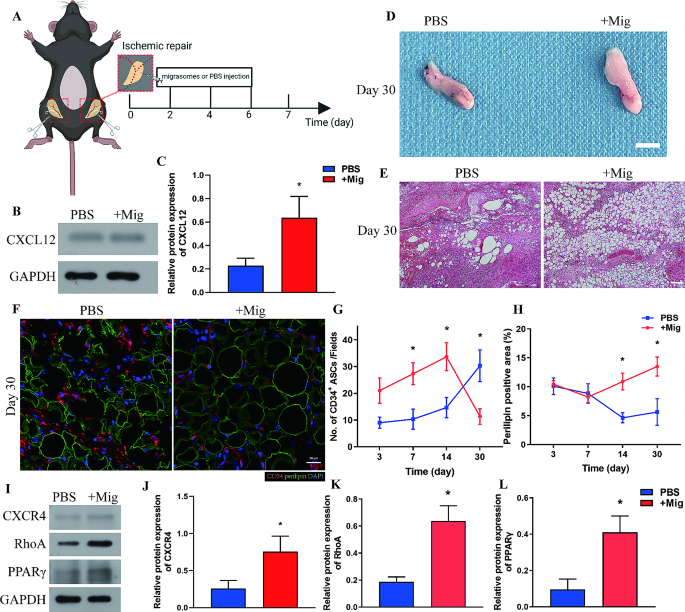
Subsequent, we investigated the position of ASC-derived migrasomes in adipose tissue regeneration by including remoted migrasomes to poorly vascularized adipose tissue post-injury. Migrasomes (+ Mig group) or phosphate-buffered saline (PBS, PBS group) have been focally injected into inguinal fats pads (Ischemic restore mannequin) subjected to the identical process because the Ischemic group for six consecutive days post-injury (Fig. 5A). Migrasomes, together with stem cell-derived migrasomes, are enriched with CXCL12 [19, 21, 22]. Persistently, western blot evaluation confirmed that expression of CXCL12 was considerably larger within the + Mig group than within the PBS group at Day 7 after harm (Fig. 5B, C). Macroscopic statement on Day 30 confirmed that tissues within the + Mig group had a extra pure look and comfortable texture whereas these within the PBS group appeared atrophic and felt inflexible (Fig. 5D). HE staining of adipose tissues on Day 30 indicated that adipose tissue regeneration was higher within the + Mig group than within the PBS group. Giant vacuoles, oil cyst-like construction, and extreme fibrosis have been noticed within the PBS group, whereas commonly organized spherical adipocytes of various sizes have been noticed within the + Mig group (Fig. 5E).
Migrasomes promote adipose tissue regeneration. (A) Schematic illustration of injection of poorly vascularized Ischemic adipose tissue with migrasomes (+ Mig group) or PBS (PBS group). N = 7 in every teams. (B) Western blot evaluation of CXCL12 protein expression in adipose tissue from the + Mig and PBS teams at 7 days after surgical procedure. (C) Semiquantitative evaluation of CXCL12 protein expression. *p < 0.05 in contrast with PBS group. (D) Macroscopic picture of adipose tissue from the + Mig and PBS teams at 30 days after surgical procedure. Scale bar, 50 mm. (E) HE staining of adipose tissue from the + Mig and PBS teams at 30 days after surgical procedure. Scale bar, 200 μm. (F) Immunofluorescence staining of CD34 and perilipin in adipose tissue from the + Mig and PBS teams at 30 days after surgical procedure. Scale bar, 20 μm. (G) Quantitative evaluation of infiltrated CD34+ ASCs per area in adipose tissue from the + Mig and PBS teams over time. (H) Quantitative evaluation of the perilipin+ space per area in adipose tissue from the + Mig and PBS teams over time. *p < 0.05 in contrast with PBS group. (I) Western blot evaluation of CXCR4, RhoA, and PPARγ protein expression in adipose tissue from the + Mig and PBS teams 7 days after surgical procedure. (J-L) Semiquantitative evaluation of CXCR4, RhoA, and PPARγ protein expression. *p < 0.05 in contrast with PBS group. The information are imply ± SEM. Statistical variations in (C) have been examined by nonparametric Mann-Whitney take a look at. (G), (H) and (J) to (L) have been analyzed by One-way ANOVA
To confirm the mechanism by which migrasomes promote tissue regeneration, immunofluorescence staining of CD34 and perilipin was carried out. Quantification of CD34+ cells confirmed that migrasomes promote the infiltration of ASCs as early as Day 3 post-injury. The variety of ASCs sharply elevated from Day 3 to Day 14 within the + Mig group however remained comparatively low within the PBS group. The variety of ASCs decreased within the + Mig group however elevated within the PBS group from Day 14 to Day 30, indicating ASCs are required for regenerative occasions in PBS group (Fig. 5F, G). The perilipin+ space remained low in each teams within the first 7 days. The perilipin+ space within the + Mig group elevated from Day 7 to Day 30. Against this, the perilipin+ space within the PBS group dropped from Day 7 to Day 14 and barely elevated to Day 30 (Fig. 5F, H). Tissue buildings remained damaged with considerably extra infiltrated ASCs within the PBS group, whereas extra full tissue buildings with fewer ASCs have been noticed within the + Mig group (Fig. 5F). In abstract, we confirmed that migrasomes can promote adipose tissue regeneration by facilitating early recruitment of ASCs.
CXCL12 binds to its receptor CXCR4 and prompts RhoA by activation of the small G proteins, Gi and Gα13, which ends up in directional cell migration [34, 35]. Western blot evaluation of tissues on Day 7 confirmed that protein expression of CXCR4 and RhoA was considerably larger within the + Mig group than within the PBS group (Fig. 5I-Ok), indicating that migrasomes activate CXCR4/RhoA signaling. Moreover, addition of migrasomes promoted expression of the adipogenesis-associated protein PPAR-γ (Fig. 5I, L). These information present that migrasomes enriched with CXCL12 promote adipose tissue regeneration by recruiting ASCs by CXCR4/RhoA signaling. The recruited ASCs increase tissue regeneration in all probability by selling adipogenesis as proven by elevated expression of PPAR-γ.
Blockade of ASC infiltration reduces the variety of migrasomes
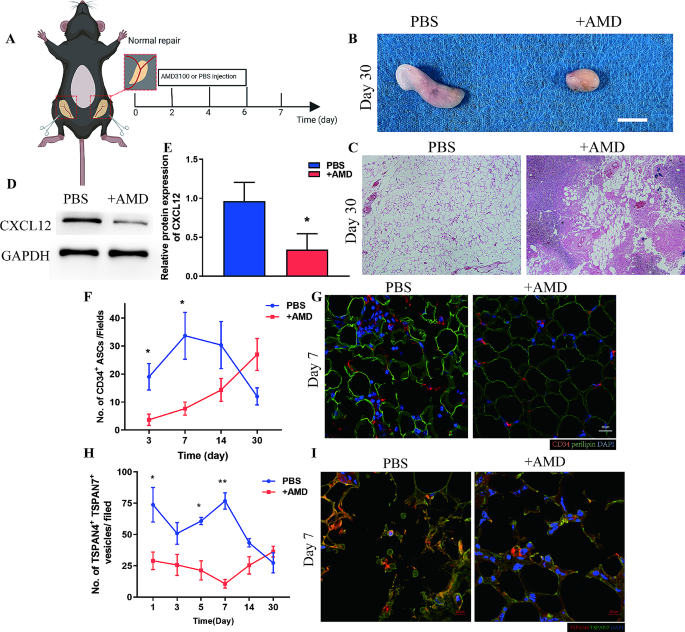
To show the origin of migrasomes in vivo, we blocked infiltration of ASCs utilizing the CXCR4 inhibitor AMD3100 [24]. AMD3100 (+ AMD group) or PBS (PBS group) was focally injected to the inguinal fats pads subjected to the identical process because the Regular group (Fig. 6A). The in vivo use of AMD3100 forestall the infiltration of ASCs, which worsen the regeneration of adipose tissue. Macroscopic statement on Day 30 put up operation confirmed adipose tissue from + AMD group with decreased tissue mass and atrophy look and brittle texture whereas that from PBS group with pure look and comfortable texture (Fig. 6B). HE staining confirmed tissue construction on Day 30 from PBS group was steady, with spherical adipocytes organized tightly and commonly whereas giant vacuoles, extreme cell infiltration with disorganized construction might be witnessed from adipose tissue of + AMD group (Fig. 6C). Moreover, western blot evaluation of tissue from Day 7 confirmed expression of CXCL12 was considerably larger within the PBS group than within the + AMD group (Fig. 6D-E). Quantitative evaluation of CD34+ cells confirmed that AMD3100 blocked infiltration of ASCs, with considerably decrease ranges of CD34+ cells within the + AMD group than within the PBS group over the primary 7 days (Fig. 6B, C). Immunofluorescence staining of TSPAN4 and TSPAN7 confirmed that blockade of ASC infiltrations considerably decreased the variety of migrasomes throughout tissue regeneration (Fig. 6D, E). These information present proof that ASCs can generate migrasomes enriched with CXCL12 to advertise ASC infiltration throughout adipose tissue regeneration.
ASCs generate migrasomes in vivo. (A) Schematic illustration of inhibition of ASC infiltration in Regular adipose tissue utilizing CXCR4 inhibitor AMD3100 (+ AMD). N = 7 in every teams. (B) Macroscopic picture of adipose tissue from the PBS and + AMD teams at 30 days after surgical procedure. Scale bar, 50 mm. (C) HE staining of adipose tissue from the PBS and + AMD teams at 30 days after surgical procedure. Scale bar, 200 μm. (D) Western blot evaluation of CXCL12 protein expression in adipose tissue from the PBS and + AMD teams at 7 days after surgical procedure. (E) Semiquantitative evaluation of CXCL12 protein expression. *p < 0.05 in contrast with PBS group. (F) Immunofluorescence staining of CD34 and perilipin in adipose tissue from the + AMD and PBS teams at 7 days after surgical procedure. Scale bar, 20 μm. (G) Quantitative evaluation of infiltrated CD34+ ASCs per area in adipose tissue from the + AMD and PBS teams over time. *p < 0.05 in contrast with PBS. (H) Immunofluorescence staining of TSPAN4 and TSPAN7 in adipose tissue from the + AMD and PBS teams at 7 days after surgical procedure. Scale bar, 10 μm. (I) Quantitative evaluation of migrasomes per area in adipose tissue from the + AMD and PBS teams over time. *p < 0.05, **p < 0.01 in contrast with PBS. The information are imply ± SEM. Statistical variations have been analyzed utilizing One-way ANOVA
Migrasomes improve migration of ASCs through CXCR4/RhoA in vitro
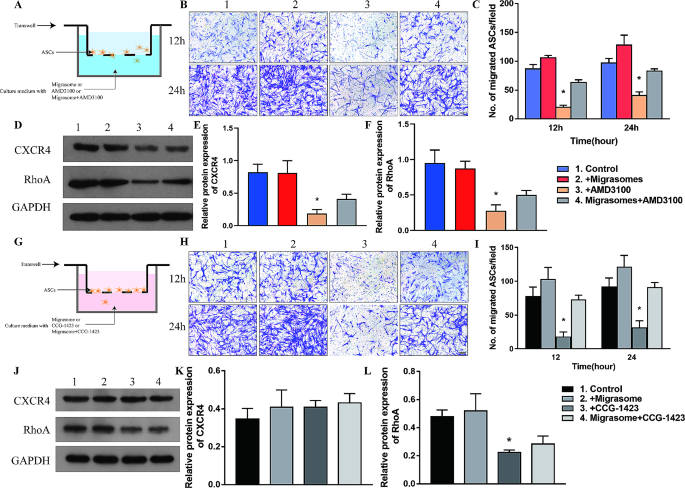
To confirm the capability of migrasomes to advertise migration of ASCs, we carried out transwell assay with migrasomes, AMD3100, and migrasomes plus AMD3100 (migrasome + AMD3100 group) (Fig. 7A). Migration capability of ASCs was enhanced by migrasomes, diminished by AMD3100, and restored within the migrasome + AMD3100 group (Fig. 7B, C). Western blot and qPCR analyses of cultured cells confirmed that expression of CXCR4 and RhoA was upregulated by migrasomes and inhibited by AMD3100 (Fig. 7D-F, Further file 6: Determine S6). Expression of CXCR4 and RhoA was partially restored within the migrasome + AMD3100 group, suggesting that migrasomes promote migration of ASCs through CXCR4/RhoA signaling.
Migrasomes promote migration of ASCs in vitro through activation of CXCR4/RhoA signaling by CXCL12. (A) Schematic illustration of the transwell assay of ASCs with the management, migrasomes, AMD3100, and Migrasome + AMD3100 teams. Information are obtained from 3 impartial reduplicated experiments. (B) Photographs of migrated ASCs after incubation for 12 and 24 h. Scale bar, 200 μm. (C) Quantitative evaluation of the variety of migrated ASCs per area. *p < 0.05 in contrast with management. (D) Western blot evaluation of CXCR4 and RhoA protein expression. (E-F) Semiquantitative evaluation of CXCR4 and RhoA protein expression. *p < 0.05 in contrast with management. (G) Schematic illustration of the transwell assay of ASCs with the management, migrasomes, CCG-1423 or Migrasomes + CCG-1423 teams. Information are obtained from 3 impartial reduplicated experiments. (H) Photographs of migrated ASCs after incubation for 12 and 24 h. Scale bar, 200 μm. (I) Quantitative evaluation of the variety of migrated ASCs per area. *p < 0.05 in contrast with management. (J) Western blot evaluation of CXCR4 and RhoA protein expression. (Ok-L) Semiquantitative evaluation of of CXCR4 and RhoA protein expression. *p < 0.05 in contrast with management. The information are imply ± SEM. Statistical variations in (C) was assessed by Kruskal-Wallis. (E), (F), (I), (Ok) and (L) have been analyzed utilizing One-way ANOVA adopted by Bonferroni posttest
The transwell assay was carried out with management, migrasomes, CCG-1423 (a RhoA inhibitor), and migrasomes plus CCG-1423 (migrasome + CCG-1423 group) (Fig. 7G). Migration of ASCs was promoted by migrasomes, diminished by CCG-1423, and restored within the migrasome + CCG-1423 group (Fig. 7H, I). Western blot and qPCR analyses confirmed that migrasomes elevated expression of RhoA, whereas CCG-1423 inhibited expression of RhoA and had no affect on expression of CXCR4 (Fig. 7J-L, Further file 7: Determine S7). Expression of RhoA was partially rescued whereas expression of CXCR4 was not considerably altered within the migrasome + CCG-1423 group (Fig. 7J-L, Determine S7). In abstract, we demonstrated that migrasomes enriched with CXCL12 promote migration of ASCs in vitro through CXCR4/RhoA signaling.