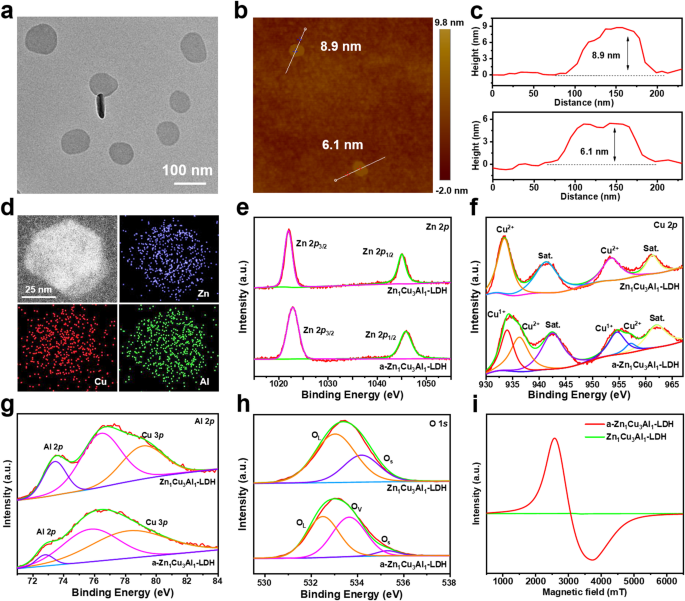
Synthesis and characterization of crystalline ZnCuAl-LDH nanoparticles
The crystalline ZnCuAl-LDH nanoparticles have been synthesized through a hydrothermal technique in keeping with a beforehand reported literature [44]. The X-ray diffraction (XRD) evaluation confirms the excessive crystallinity of the as-synthesized ZnCuAl-LDH nanoparticles (Fig. S1, purple line), with diffraction peaks at 2θ = 10.48° (003 aircraft) and 21.08° (006 aircraft), attribute of LDH constructions. The high-resolution transmission electron microscopy (HRTEM) picture (Fig. 1a) shows a two-dimensional (2D) nanoparticle morphology with a dimension of 70–120 nm and a lattice fringes of 0.383 nm (Fig. S2a), aligning with the (006) crystallographic aircraft. The chosen space electron diffraction (SAED) sample (Fig. S2b) additional confirms the single-crystalline function of ZnCuAl-LDH nanoparticles via hexagonally organized diffraction spots. Atomic pressure microscopy (AFM) evaluation reveals that the thickness of ZnCuAl-LDH nanoparticles is 6–9 nm (Fig. 1b, c). Vitality-dispersive X-ray spectroscopy (EDX) elemental mappings display uniform distribution of Zn, Cu, and Al throughout the nanoparticles (Fig. 1d), whereas inductively coupled plasma-atomic emission spectrometry (ICP-AES) quantification offers stoichiometric validation of the ternary steel composition (Desk S1).
Synthesis and characterization of LDH nanoparticles. (a) HRTEM picture of LDH nanoparticles. (b) AFM picture of LDH nanoparticles and (c) corresponding top profiles. (d) EDX mapping photographs of LDH nanoparticles. XPS (e) Zn 2p, (f) Cu 2p, (g) Al 2p, and (h) O 1s spectra of LDH and a-LDH nanoparticles. (i) ESR spectra of LDH and a-LDH nanoparticles
The chemical composition of ZnCuAl-LDH nanoparticles was analyzed utilizing X-ray photoelectron spectroscopy (XPS). Within the Zn 2p spectral area (Fig. 1e), attribute spin-orbit doublets have been noticed with binding energies at 1022.1 eV (Zn2+ 2p3/2) and 1045.1 eV (Zn2+ 2p1/2), confirming the presence of divalent zinc species. The Cu 2p spectrum (Fig. 1f) displays two distinct peaks at 933.5 eV (2p3/2) and 953.6 eV (2p1/2), in step with the oxidation state of copper(II). Spectral overlap between Al 2p and Cu 3p areas was resolved via peak deconvolution, revealing a predominant Al3+ sign at 73.0 eV together with secondary Cu2+ contributions at 77.1 eV and 79.6 eV (Fig. 1g). Oxygen speciation evaluation demonstrates two oxygen environments within the O 1s spectrum: a dominant lattice oxygen part (OL) at 533.0 eV and a minor surface-adsorbed oxygen species (OS) at 534.2 eV (Fig. 1h).
Crystalline-to-amorphous section engineering through acid etching
To induce crystalline-to-amorphous section engineering, the ZnCuAl-LDH nanoparticles underwent acidic etching in PBS 4.0, yielding amorphous ZnCuAl-LDH nanoparticles (a-ZnCuAl-LDH). Complete characterization utilizing XRD, HRTEM, XPS, Electron spin resonance (ESR) spectroscopy, zeta potential, and DLS analyses elucidated morphological and compositional alterations post-etching. XRD sample of a-ZnCuAl-LDH displays a featureless profile (Fig. S1, orange line), confirming the lack of crystallinity with amorphous construction. HRTEM photographs (Fig. S3) reveal the retention of the nanoparticle morphology with a dimension of 60–110 nm together with a disappeared lattice fringes and decreased Zn, Cu, and Al content material (Desk S1), indicating that partial steel ions have been etched away. The XPS evaluation (Fig. 1e-h) demonstrates retained Zn2+ (Zn 2p: 1022.1/1045.1 eV) and Al3+ (Al 2p: 73.0 eV) binding energies, whereas Cu 2p deconvolution reveals peaks at 934.0 eV (2p3/2) and 954.6 eV (2p1/2), alongside an O 1s part at 533.6 eV. These shifts signify Cu+ formation and OVs era, corroborating redox-active etching results. ESR spectroscopy additional confirms the structural defects in a-ZnCuAl-LDH, as evidenced by a outstanding resonance sign at G = 2.2 (Fig. 1i), absent in pristine ZnCuAl-LDH. Floor cost inversion was noticed post-etching (Fig. S4a), with zeta potentials shifting from + 44.4 ± 1.5 mV (ZnCuAl-LDH) to − 5.9 ± 1.1 mV (a-ZnCuAl-LDH). Concurrently, hydrodynamic diameter decreased from 109.7 ± 6.4 nm to 90.6 ± 8.3 nm (Fig. S4b), reflecting decreased zeta potential and hydrodynamic dimension brought on by acid etching.
Radical scavenging efficiency analysis
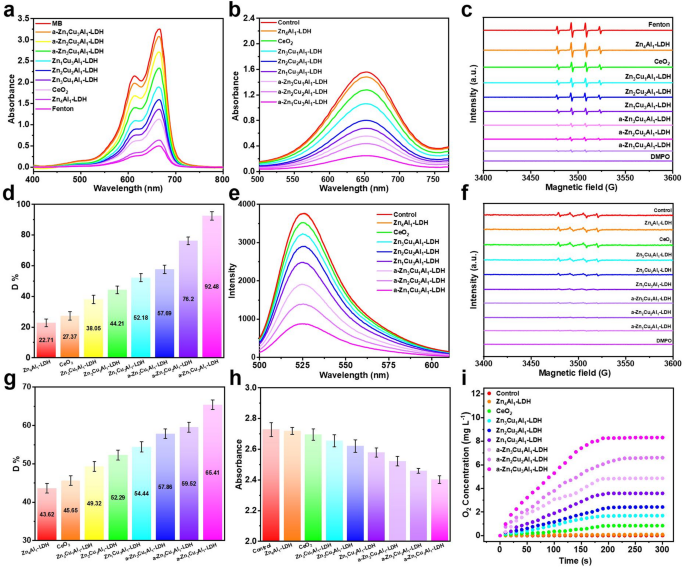
The ROS scavenging capacities of ZnCuAl-LDH and a-ZnCuAl-LDH with completely different Cu content material have been systematically evaluated. Methylene blue (MB) degradation assays beneath Fenton response situation (Fe2+/H2O2) revealed distinct ·OH scavenging behaviors of ZnCuAl-LDH and a-ZnCuAl-LDH. In management system missing scavengers, fast MB degradation occurred beneath Fe2+/H2O2 situation, as evidenced by pronounced absorbance discount (Fig. 2a). Introduction of Zn4− xCuxAl1-LDH and a-Zn4− xCuxAl1-LDH (x = 1, 2, 3) attenuated MB decomposition charges, with a-Zn4− xCuxAl1-LDH samples demonstrating superior inhibition efficacy, manifested as minimal absorbance decline relative to pristine counterparts. Notably, undoped ZnAl-LDH exhibited negligible ·OH interception, whereas Cu-rich compositions achieved vital MB preservation, presumably because of Cu+/Cu2+ redox cycle-mediated ROS scavenging. Particularly, the degradation of MB was considerably inhibited with the rise of Cu doping content material, underscoring Cu’s crucial position in radical scavenging. Comparative evaluation recognized each LDH techniques as outperforming CeO2 nanoparticles (a generally used ROS scavenger) in ·OH neutralization effectivity. Complementary verification through 3,3’,5,5’-tetramethylbenzidine (TMB) assays confirmed marked attenuation of oxidation-induced absorbance in a-Zn4− xCuxAl1-LDH techniques (Fig. 2b and S5), in step with MB assay developments. ESR spectral evaluation additional corroborated these findings, with a-Zn4− xCuxAl1-LDH samples exhibiting distinct sign attenuation at attribute ·OH adduct positions (Fig. 2c). Quantitative evaluation utilizing a ·OH assay equipment confirmed Cu-dependent ·OH suppression, the place a-Zn1Cu3Al1-LDH achieved the best inhibition price of ·OH (~ 92.5%), considerably exceeding that of different teams (Fig. 2d), which is 3.38 occasions that of CeO2.
UV-vis absorption spectra of (a) MB, (b) TMB, and (c) ESR spectra indicating ·OH scavenging within the presence of ZnAl-LDH, CeO2, Zn4− xCuxAl1-LDH, and a-Zn4− xCuxAl1-LDH (x = 1, 2, 3). (d) ·OH scavenging actions of ZnAl-LDH, CeO2, Zn4− xCuxAl1-LDH, and a-Zn4− xCuxAl1-LDH by ·OH Assay Equipment. (e) Fluorescence spectra of DHR123 and (f) ESR spectra for evaluating ·O2− scavenging within the presence of ZnAl-LDH, CeO2, Zn4− xCuxAl1-LDH, and a-Zn4− xCuxAl1-LDH. (g) ·O2− scavenging actions of ZnAl-LDH, CeO2, Zn4− xCuxAl1-LDH, and a-Zn4− xCuxAl1-LDH by SOD Assay Equipment. (h) Absorbance of H2O2 at 240 nm and (i) O2 era curves within the presence of ZnAl-LDH, CeO2, Zn4− xCuxAl1-LDH, and a-Zn4− xCuxAl1-LDH
Constructing on ·OH scavenging research, the ·O2– scavenging efficacy (SOD-like exercise: the disproportionation of ·O2– to H2O2 and O2) of a-Zn4− xCuxAl1-LDH was probed via complementary methodologies. Dihydroethidium (DHE) and dihydrorhodamine 123 (DHR 123) fluorescence detection, delicate to ·O2– era in xanthine/xanthine oxidase techniques, revealed distinct scavenging behaviors. Whereas management and ZnAl-LDH teams exhibited maximal fluorescence depth (Fig. S6, S7 and Fig. 2e), CeO2 and Zn4− xCuxAl1-LDH teams confirmed average sign discount. Notably, a-Zn4− xCuxAl1-LDH teams induced markedly enhanced fluorescence attenuation, with a-Zn1Cu3Al1-LDH attaining probably the most pronounced quenching, in step with its superior ·OH scavenging capability. ESR spectroscopy utilizing 5,5-dimethyl-1-pyrroline N-oxide (DMPO) spin traps corroborated these developments (Fig. 2f). a-Zn4− xCuxAl1-LDH techniques demonstrated pronounced suppression of ·O2–-specific spectral signatures in comparison with the management, ZnAl-LDH, CeO2, and Zn4− xCuxAl1-LDH teams, with a-Zn1Cu3Al1-LDH once more exhibiting maximal sign diminution. Quantitative evaluation through SOD Assay Equipment additional validated Cu-dependent ·O2– scavenging (Fig. 2g), with a-Zn1Cu3Al1-LDH attaining peak effectivity (65.4%), which is 1.43 occasions that of CeO2.
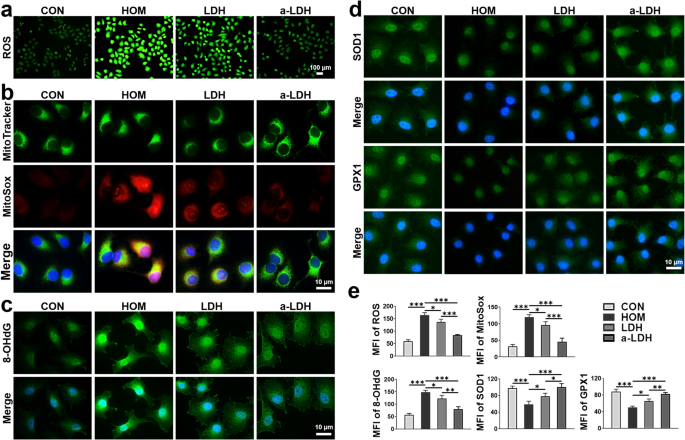
a-LDH mimics enzyme exercise to scavenge extra ROS and inhibit oxidative injury. (a) Consultant ROS photographs in HOM-cultured (500 mOsM) HCE-2 cells after therapy with 2 µg mL−1 LDH and a-LDH by DCFH-DA staining. (b) Consultant MitoSox, (c) 8-OHdG, and (d) SOD1 and GPX1 immunofluorescence staining photographs in HCE-2 cells after indicated remedies beneath oxidative stress. (e) Quantitative evaluation of the imply fluorescence depth (MFI) of ROS, MitoSox, 8-OHdG, SOD1, and GPX1. Information are offered as imply values ± SD. (n = 3). *p < 0.05, **p < 0.01, and ***p < 0.001
Given the propensity of ·O2– scavenging to generate H2O2, the CAT-like exercise of a-Zn4− xCuxAl1-LDH was additionally assessed by monitoring UV-vis spectral adjustments at 240 nm, attribute of H2O2 decomposition. In Fig. 2h, i, a-Zn4− xCuxAl1-LDH induced marked attenuation of the 240 nm absorbance sign alongside visible statement of oxygen effervescence in contrast with different teams, confirming catalytic H2O2 dissociation. Furthermore, a Cu-dependent enhancement of CAT-like exercise was noticed, with a-Zn1Cu3Al1-LDH demonstrating probably the most vital H2O2 decomposition kinetics. This cascade impact highlights the twin performance of a-Zn1Cu3Al1-LDH: ·O2– scavenging coupled with in situ H2O2 cleansing through redox-active copper facilities. Collectively, these multimodal assessments set up a-Zn1Cu3Al1-LDH because the optimum ROS scavenger, prompting its choice for subsequent research (until in any other case specified, a-Zn1Cu3Al1-LDH is denoted as a-LDH).
Mechanistic insights
To elucidate the mechanistic foundation of a-LDH’s ROS scavenging capability, XPS valence state evaluation was carried out pre- and post-ROS publicity. XPS Zn 2p and Al 2p spectra of post-a-LDH maintained binding vitality profiles an identical to pre-a-LDH (Fig. S8a, b), confirming structural integrity of those metallic facilities. Crucially, XPS Cu 2p spectra (Fig. S8c) revealed sustained Cu+/Cu2+ redox biking in post-a-LDH, suggesting dynamic electron alternate between oxidation states, a crucial facilitator of ROS scavenging. Oxygen speciation evaluation (XPS O 1s) demonstrated substantial attenuation of OV-associated spectral options in post-a-LDH (Fig. S8d), indicative of vacancy-mediated ROS adsorption and subsequent redox-mediated ROS scavenging. These findings collectively set up a twin pathway scavenging mechanism: Persistent Cu+/Cu2+ redox {couples} allow electron shuttling for radical disproportionation, whereas OVs act as adsorption websites and reactive facilities for ROS decomposition. The synergistic interplay between redox-active copper species and defect engineering underpins antioxidative efficiency enhancement of a-LDH.
Floor functionalization for enhanced stability
To optimize dispersion stability and biocompatibility, each ZnCuAl-LDH and a-ZnCuAl-LDH nanoparticles have been functionalized with QCS-PBA and PEG (mQCS−PBA: mPEG=molar ratio 1:0.2), yielding modified LDH and a-LDH composites. The PEG part imparted steric stabilization via its hydrophilic polymer chains, mitigating nanoparticle aggregation, whereas QCS-PBA enhanced colloidal stability by forming dynamic boron ester bonds between phenylboronic acid moieties in QCS-PBA and floor hydroxyl teams in LDH. Fourier remodel infrared (FT-IR) spectroscopy confirmed profitable floor modification, with modified LDH and a-LDH exhibiting attribute vibrational modes at 2877 cm−1 (C-O-C stretching of PEG), 1641 cm−1 (C = N/C-O stretching vibration in amide II band and C-O stretching vibration in acetyl group of QCS-PBA), and 1384 cm−1 (NO3− N-O vibration of LDH) (Fig. S9). Dynamic mild scattering (DLS) measurements corroborated functionalization efficacy, displaying zeta potential shifts to + 46.5 ± 2.1 mV (modified LDH) and + 43.0 ± 1.8 mV (modified a-LDH) in comparison with unmodified counterparts (Fig. S4a). DLS evaluation additionally revealed modest hydrodynamic dimension increments post-modification (modified LDH: 123.6 ± 7.7 nm; modified a-LDH: 114.3 ± 5.8 nm; Fig. S4b), with each composites sustaining dispersion stability over 2 days (Fig. S10). The pictures of composites in aqueous resolution at varied time factors visually validated the improved dispersion conferred by QCS-PBA-PEG modification (Fig. S11).
The antioxidative skill of a-LDH in vitro
Initially, the viability of human corneal epithelial cells (HCE-2) and conjunctival epithelial cells (CCL-20.2) handled with concentration-gradient LDH or a-LDH was analyzed utilizing the cell counting kit-8 (CCK-8) assay. Outcomes confirmed that each LDH and a-LDH maintained cell viability above 80% in HCE-2 and CCL-20.2 cells at a focus of 250 µg mL−1, indicating wonderful biocompatibility of those supplies (Fig. S12a, b). Subsequently, to find out the optimum motion focus of LDH and a-LDH for follow-up experiments, the viability of HCE-2 cells was investigated beneath a 500 mOsM hyperosmotic mannequin (HOM) that successfully mimics the dry eye microenvironment by potentiating ROS manufacturing and inflammatory responses [51]. The outcomes indicated that pretreatment with 2 µg mL−1 a-LDH considerably elevated the typical viability of HCE-2 cells from 53% to 65% beneath hypertonic circumstances (Fig. S13). In distinction, therapy with 2 µg mL−1 LDH alone solely enhanced cell viability to 55%, with out statistical significance. Additional growing the focus of a-LDH to 10 µg mL−1 didn’t yield extra enhancements in cell survival beneath hypertonic stress. This outcome means that the protecting impact of a-LDH in opposition to hypertonicity-induced oxidative injury could not comply with a concentration-dependent sample. Due to this fact, 2 µg mL−1 was chosen because the optimum focus for subsequent experiments.
Oxidative stress performs a pivotal position in DED pathogenesis, involving elevated intracellular ROS ranges that induce injury to lipids, proteins and DNA [52]. The antioxidant actions of a-LDH in opposition to intracellular ROS have been assessed by DCFH-DA fluorescence staining. The depth of inexperienced fluorescence corresponds to ROS ranges. As proven in Fig. 3a, following hypertonic stimulation, a sturdy inexperienced fluorescence sign was noticed within the HOM group, which was considerably stronger than that of the management (CON) group, indicating elevated intracellular ROS ranges. Comparatively, the imply fluorescence depth (MFI) decreased within the LDH group (P < 0.05) (Fig. 3e). Notably, the a-LDH handled group confirmed the weakest MFI in comparison with the HOM and LDH teams (p < 0.001), demonstrating its superior ROS scavenging skill.
Moreover, as mitochondria represent the first supply of intracellular ROS, focused scavenging of mitochondrial ROS (mtROS) can restore mitochondrial redox homeostasis, thereby assuaging DED [53, 54]. Due to this fact, it’s important to find out the mtROS scavenging exercise of a-LDH. MitoSox was used as a particular indicator of mtROS, with elevated mtROS ranges evidenced by intense purple fluorescence. As proven in Fig. 3b, the purple fluorescence of mtROS was remarkedly elevated within the HOM group in comparison with the management group. Each LDH and a-LDH considerably decreased mtROS ranges in HCE-2 cells uncovered to hypertonic stress. Notably, the fluorescence depth within the a-LDH group was considerably weaker than within the LDH group (Fig. 3e), with statistical significance (p < 0.001). These outcomes confirmed the numerous skill of a-LDH in suppressing pathological ROS overproduction. Moreover, the inhibitory impact of a-LDH on DNA oxidation was evaluated utilizing 8-OHdG, a biomarker of oxidative injury to DNA by ROS [55]. In Fig. 3c and e, HOM induced substantial DNA oxidation, as evidenced by intense inexperienced fluorescence. LDH attenuated the inexperienced fluorescence sign of 8-OHdG, indicating a sure impact on stopping DNA oxidation. Nonetheless, a-LDH considerably decreased the 8-OHdG degree beneath oxidative stress, exerting a superior safety impact over the LDH on suppression of DNA oxidation.
Oxidative stress is brought on by an imbalance between ROS manufacturing and elimination. Antioxidant enzymes resembling SOD and GPX1 are the first-line antioxidant protection system in organisms in opposition to the dangerous results of ROS [56, 57]. The expression ranges of SOD1 and GPX1 in HCE-2 cells have been decided by immunofluorescence. As depicted in Fig. 3d and e, in contrast with the HOM group, the fluorescence intensities of SOD1 and GPX1 considerably elevated after pretreatment with LDH and a-LDH, indicating that LDH and a-LDH scavenge extra ROS to keep up redox homeostasis by selling the excessive expression of those antioxidant enzymes. Notably, a-LDH exhibited a stronger antioxidative stress impact than LDH, presumably attributed to the enriched OVs in a-LDH enhancing the adsorption of ROS species and selling subsequent catalytic processes (Fig. 3e). These findings reveal that a-LDH possesses potent enzyme-like exercise to successfully scavenge extra ROS and inhibit oxidative injury.
Identification of the therapeutic targets of a-LDH through RNA sequencing evaluation
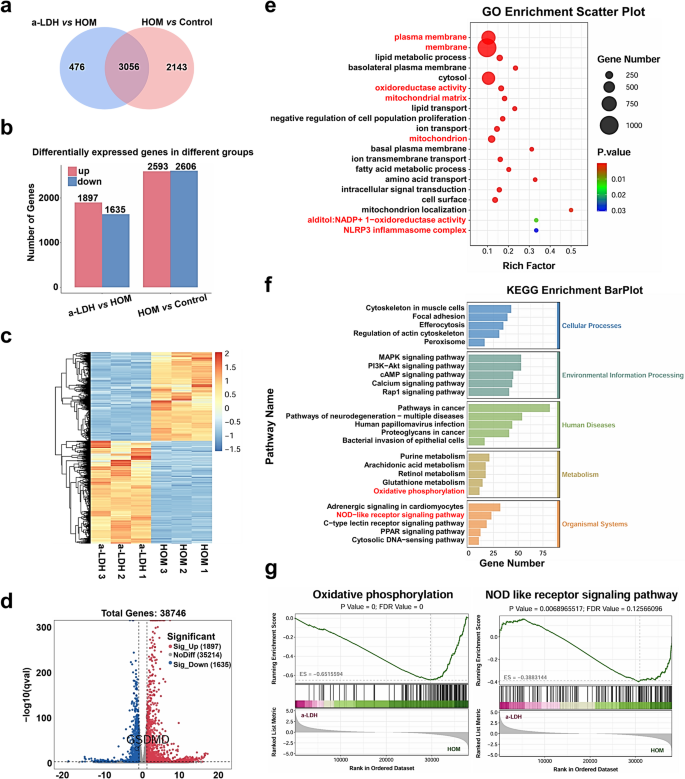
The aforementioned experiments advised that a-LDH might successfully scavenge extra ROS, however the molecular mechanisms underlying its downstream concentrating on of inflammatory pathways stay unclear. To discover the anti-inflammatory mechanisms of a-LDH in HCE-2 cells, we carried out RNA sequencing (RNA-seq) evaluation on HOM-induced HCE-2 cells. Using the standards of |log2FC| ≥ 1 and q < 0.05, a considerable variety of differentially expressed genes (DEGs) have been recognized among the many three teams (Fig. 4a, b). The comparability ‘HOM vs Management’ indicated 5199 DEGs (3056 + 2143) in HOM-treated cells in comparison with the management group (Fig. 4a). In the meantime, the comparability ‘a-LDH vs HOM’ recognized DEGs in a-LDH co-treated HOM cells relative to HOM alone with 1635 downregulated and 1897 upregulated genes (Fig. 4b). Heatmap visualization confirmed the excessive repeatability of DEGs inside teams (Fig. 4c). In line with gene ontology (GO) evaluation, a-LDH dramatically affected DEGs related to mobile membrane regulation, mitochondrial matrix, and NLRP3 inflammasome complicated (Fig. 4e). Moreover, Kyoto Encyclopedia of Genes and Genomes (KEGG) evaluation demonstrated that DEGs have been considerably enriched in oxidative phosphorylation and NOD-like receptor signaling pathways (Fig. 4f). Notably, the ROS are thought-about as dangerous by-products of mitochondrial metabolism, particularly oxidative phosphorylation [58]. Gene Set Enrichment Evaluation (GSEA) confirmed that a-LDH downregulated oxidative phosphorylation and NOD-like receptor signaling pathways (Fig. 4g). Moreover, a-LDH down-regulated GSDMD in DEGs (Fig. 4d), which mediates inflammatory cytokine launch and mobile pyroptosis via the NLRP3 signaling pathway. Primarily based on these findings, we chosen NLRP3/Caspase-1/GSDMD signal-mediated pyroptosis regulation as a candidate goal for additional investigation.
RNA-seq evaluation of therapeutic targets of a-LDH on HOM-induced HCE-2 cells. (a) Completely different expression genes (DEGs) are frequent among the many HOM vs Management and a-LDH vs HOM. (b) The variety of DEGs. (c) Heatmap and (d) Volcano plot of the DEGs. Crimson dots denote upregulated genes; blue dots denote downregulated genes. (e) Gene Ontology (GO) enrichment evaluation between a-LDH vs HOM. Various colours denote statistical significance, whereas the dimensions of every circle corresponds to the variety of genes. (f) KEGG evaluation of DEGs between a-LDH vs HOM. (g) Oxidative phosphorylation and NOD like receptor signaling pathways primarily based on GSEA
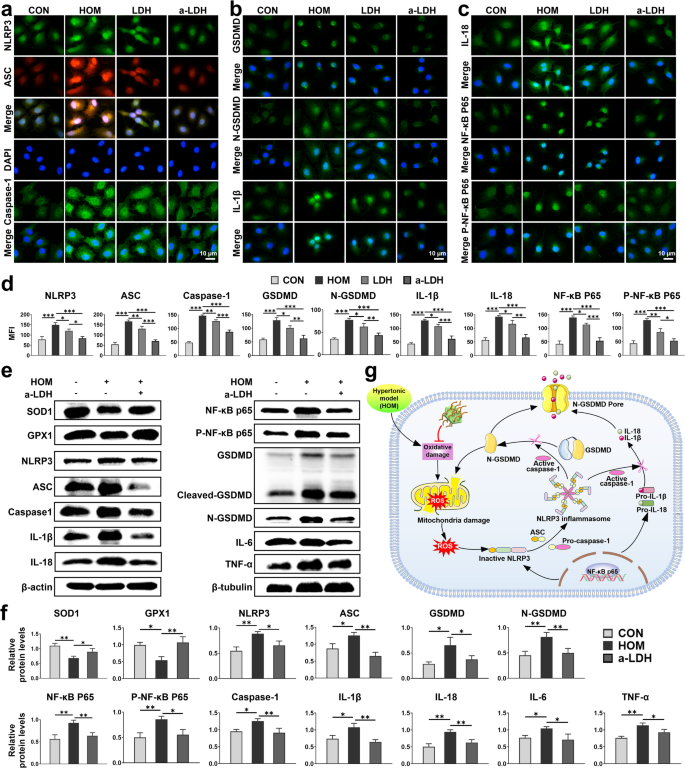
NLRP3/Caspase-1/GSDMD-mediated pyroptosis inhibition in vitro
NLRP3/Caspase-1/GSDMD-mediated pyroptosis pathway considerably contributes to the pathogenesis of DED by selling the discharge of pro-inflammatory cytokines and exacerbating ocular floor injury [23, 59]. Thus, we investigated the results of LDH and a-LDH on hyperosmolarity-induced pyroptosis in HCE-2 cells by immunofluorescence assays. The outcomes revealed that the expression ranges of NLRP3, ASC, Caspase-1, GSDMD, N-GSDMD, IL-1β, IL-18, NF-κB P65, P-NF-κB P65, TNF-α, and IL-6 within the a-LDH group have been considerably decrease than these within the HOM group (Fig. 5a-c, Fig. S14), with statistical significance confirmed (Fig. 5d, p < 0.001). Furthermore, a-LDH exhibited superior efficacy to LDH (p < 0.05). These findings indicated that a-LDH exerted a extra useful impact on assuaging elevated ROS ranges and inhibiting NLRP3 inflammasome activation-induced pyroptosis brought on by oxidative stress. To additional validate the antioxidant and anti-pyroptotic results of a-LDH, western blot evaluation was carried out (Fig. 5e). The outcomes demonstrated that a-LDH therapy dramatically decreased the degrees of hyperosmolarity-induced pro-inflammatory markers, together with NLRP3, ASC, Caspase-1, GSDMD, N-GSDMD, IL-1β, IL-18, TNF-α, and IL-6, whereas concurrently upregulating the expression of antioxidant enzymes SOD1 and GPX1 (Fig. 5e, f, p < 0.05). These knowledge collectively spotlight the therapeutic potential of a-LDH in mitigating pyroptosis via assuaging oxidative stress and inhibiting the downstream NLRP3/Caspase-1/GSDMD pathway (Fig. 5g).
NLRP3/Caspase-1/GSDMD-mediated pyroptosis inhibition in HCE-2 cells. Immunofluorescence staining photographs of (a) NLRP3, ASC, Caspase-1, (b) GSDMD, N-GSDMD, IL-1β, (c) IL-18, NF-κB P65, P-NF-κB P65, and (d) corresponding quantitative evaluation of MFI. (e) Proteins concerned within the NLRP3/Caspase-1/GSDMD signaling pathway have been examined by western blot, and (f) quantification evaluation. Information are offered as imply values ± SD. (n = 3). (g) Schematic illustration of a-LDH attenuating HOM-induced oxidative stress and inhibiting pyroptosis mediated by the downstream NLRP3/Caspase-1/GSDMD pathway. *p < 0.05, **p < 0.01, and ***p < 0.001
Mitochondrial injury alleviation mediated by a-LDH in vitro
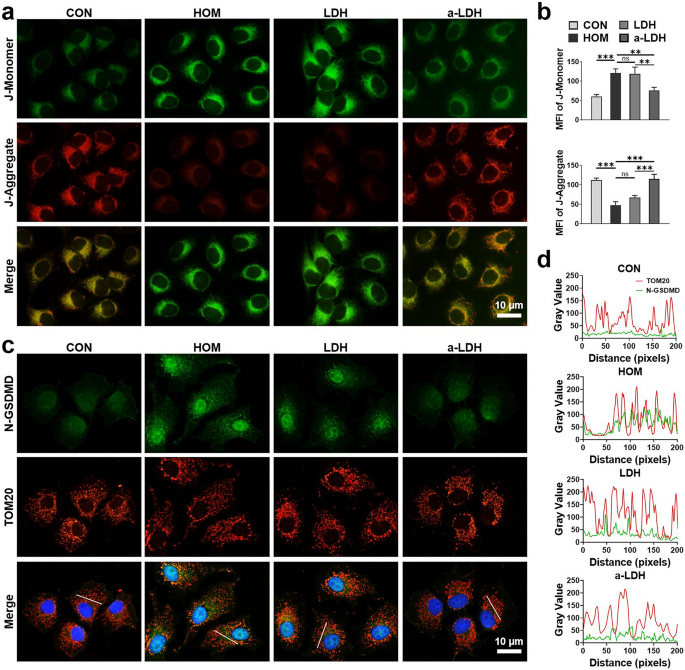
The upkeep of regular mitochondrial perform is contingent upon the preservation of mitochondrial membrane potential (ΔΨm) [60]. Disruptions on this potential are strongly linked to mitochondrial pathologies. To analyze the alterations in ΔΨm of HCE-2 cells beneath oxidative stress, we utilized the JC-1 assay equipment, which screens the fluorescence shifts from inexperienced (depolarized mitochondria) to purple (polarized mitochondria). As proven in Fig. 6a, the HOM group exhibited marked ΔΨm depolarization, characterised by intense inexperienced fluorescence. LDH therapy failed to enhance ΔΨm, as evidenced by persistently weak purple fluorescence. In distinction, a-LDH therapy demonstrated a major restorative impact on ΔΨm, with enhanced purple fluorescence indicating improved mitochondrial polarization (Fig. 6b). The outcome highlighted the potential of a-LDH to revive ΔΨm and mitochondrial perform beneath oxidative stress.
Mitochondrial injury alleviation mediated by a-LDH in vitro. (a) Consultant fluorescence photographs of JC-1 monomers and JC-1 aggregates in HCE-2 cells after therapy with LDH and a-LDH at 2 µg mL−1 and (b) corresponding quantitative evaluation of MFI. (c) Immunofluorescence co-localization evaluation of N-GSDMD and mitochondrial outer membrane protein marker TOM20 and (d) corresponding plots of pixel depth alongside the white line. Information are offered as imply values ± SD. (n = 3). *p < 0.05, **p < 0.01, ***p < 0.001, and ns p > 0.05
Hyperosmotic publicity triggers pyroptosis by activating the ROS/NLRP3/caspase-1 signaling pathway, thereby upregulating GSDMD cleavage to generate pore-forming GSDMD-N fragment [61, 62]. Accumulating proof indicated that mitochondrial oxidative stress induced by mtROS overproduction can depolarize ΔΨm and recruit N-GSDMD to the mitochondrial membranes [29, 63, 64]. Subsequently, mtROS was launched into the cytoplasm via N-GSDMD pores, the place it additional activated inflammasome complexes, thereby amplifying inflammatory responses and selling pyroptosis. This cascade highlighted the interaction between pyroptosis and mitochondrial dysfunction in driving inflammatory signaling [30, 65]. To validate the involvement of NLRP3/Caspase-1/GSDMD signaling-mediated pyroptosis in mitochondrial injury, we performed a co-localization evaluation of N-GSDMD and mitochondria utilizing TOM20, a mitochondrial outer membrane protein marker. Within the HOM and LDH teams, N-GSDMD (inexperienced fluorescence) principally overlapped with mitochondrial protein TOM20 (purple fluorescence), indicating predominant mitochondrial localization of N-GSDMD and consequent mitochondrial injury (Fig. 6c). In distinction, the a-LDH group confirmed considerably decreased expression degree of N-GSDMD in contrast with HOM and LDH. Moreover, the 2 indicators have been extremely overlapping on the pixel degree within the HOM and LDH teams, however minimal N-GSDMD was detected on mitochondrial membranes within the a-LDH group (Fig. 6d). These findings implicated a-LDH-mediated inhibition of pyroptosis contributes to assuaging mitochondrial injury and sustaining regular mitochondrial perform.
Therapeutic impact of a-LDH Wants in vivo
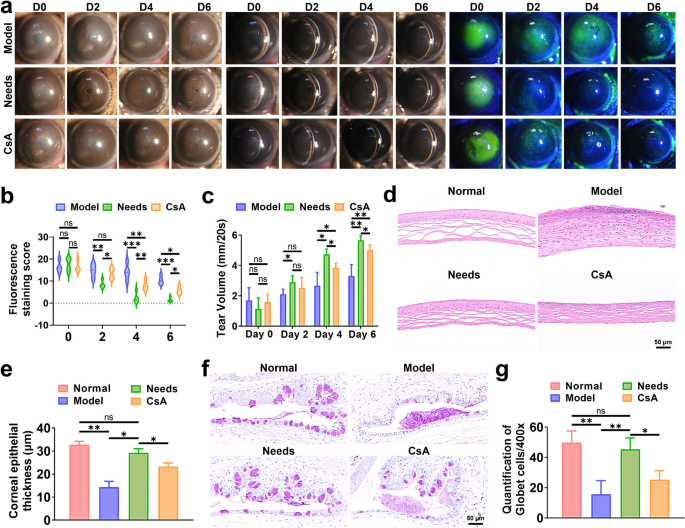
In medical follow, fluorescein staining and tear secretion experiments are generally employed as diagnostic instruments for evaluating severity of DED [66]. Due to this fact, we utilized these exams to evaluate the effectiveness of Wants in a DED mouse mannequin. DED causes numerous corneal epithelial accidents, with inexperienced fluorescence highlighting broken websites. To quantify ocular floor restore, a 25-point grading system was utilized for fluorescence staining (Fig. 7a, b). On Day 2, each the saline and cyclosporine A (CsA) teams exhibited excessive epithelial staining scores higher than 13 (14.75 ± 2.99 and 13.50 ± 2.38, respectively), suggesting persistent extreme epithelial harm. In distinction, the Wants group exhibited the bottom rating (8.50 ± 1.73). On Day 4, epithelial staining scores remained higher than 10 within the saline group, whereas that of the CsA group decreased (rating: 8.00 ± 2.16), indicating a slight enchancment in corneal epithelial harm. Remarkably, the Wants group achieved an additional decreased rating (2.50 ± 2.38), highlighting its superior skill in selling epithelial restore. However, full therapeutic of the epithelium was not but achieved. On Day 6, the saline group maintained a rating >8, reflecting unresolved epithelial injury. The CsA group had a rating of 5.75 ± 1.71, with a small quantity of fluorescence nonetheless noticed within the corneal epithelium. Notably, the Wants group displayed minimal corneal fluorescence, which grew to become nearly invisible. Schirmer take a look at outcomes confirmed that the typical tear quantity of the saline group barely raised from 1.70 ± 0.75 mm to three.30 ± 0.68 mm, indicating restricted alleviation of dryness (Fig. 7c). CsA therapy partially restored tear secretion (from 1.58 ± 0.46 to five.02 ± 0.30 mm), whereas Wants administration exhibited superior effectiveness in restoring tear secretion (from 1.14 ± 0.65 mm to five.66 ± 0.28 mm), reaching ranges akin to wholesome controls.
Therapeutic impact of Wants in vivo. (a) Optical, slit-lamp, and corneal fluorescein sodium staining micrographs, (b) fluorescein staining scores, and (c) tear quantity of the DED mice after 6 days of therapy. (d) H&E staining photographs of the cornea and (e) quantitative evaluation of the corneal epithelial thickness. (f) Consultant conjunctival PAS staining photographs and (g) the corresponding counts of goblet cells per area. Information are offered as imply values ± SD.(n = 3). *p < 0.05, **p < 0.01, ***p < 0.001 and ns p > 0.05
Within the Regular group, the corneal epithelium remained intact, well-organized, and composed of 5–6 layers of epithelial cells. In distinction, the DED group exhibited superficial epithelium detachment and irregularities, with disorganized and markedly thinned epithelial cells accompanied by in depth inflammatory cell infiltration (Fig. 7d). CsA therapy promoted corneal epithelial restoration and mitigated irritation, nevertheless, the corneal epithelium failed to revive passable regular thickness, and the corneal stroma remained densely packed. Notably, the Wants-treated group demonstrated a well-aligned epithelial construction with absolutely restored thickness (Fig. 7e). Periodic acid-Schiff (PAS) staining revealed a major lower within the variety of conjunctival goblet cells within the DED group in comparison with the conventional group. Within the CsA-treated group, goblet cell numbers steadily elevated (Fig. 7f), whereas the Wants-treated group achieved near-normal ranges of conjunctival goblet cells (Fig. 7g). These findings collectively highlighted the crucial position of Wants in repairing DED-associated corneal epithelial and conjunctival goblet cell injury.
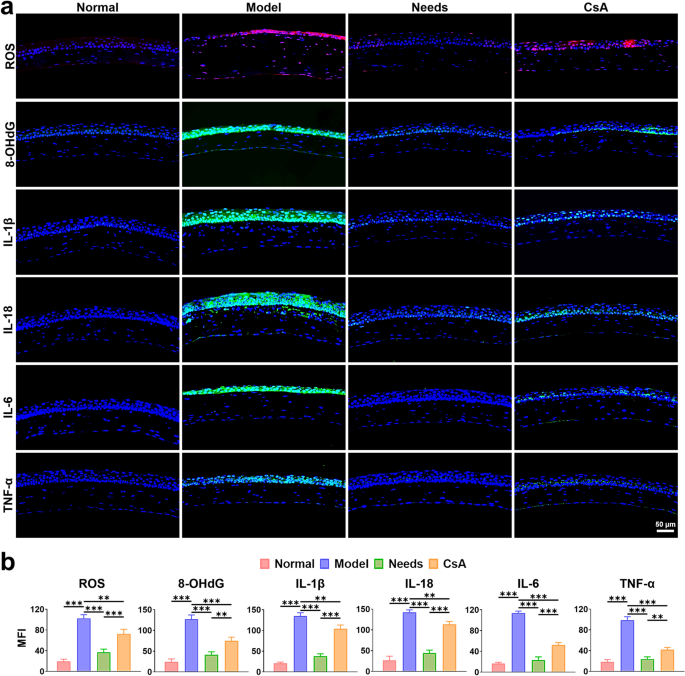
To additional discover the therapeutic potential of Wants, its anti-oxidant and anti inflammatory results have been evaluated by corneal immunofluorescence staining (Fig. 8a). The outcomes confirmed that the depth of ROS, 8-OHdG, IL-1β, IL-18, IL-6, and TNF-α staining within the DED group was considerably larger than these within the regular group. CsA decreased the expression ranges of those indicators within the cornea, whereas Wants exhibited probably the most potent suppression of their expression relative to the DED group (Fig. 8b). Taken collectively, Wants successfully repaired corneal epithelial defects, elevated conjunctival goblet cells, enhanced tear movie stability, restored tear secretion, and downregulated inflammatory cytokine expression within the DED mouse mannequin, demonstrating its outstanding therapeutic efficacy.
The antioxidant and anti inflammatory results of Wants in vivo. (a) Analysis of oxidative stress indicators (ROS, 8-OHdG, inflammatory elements IL-1β, IL-18, IL-6, and TNF-α) by immunofluorescence staining on the corneal epithelium within the regular and DED mice eyes following topical utility of saline, Wants, CsA. (b) Quantitative evaluation of MFI of ROS, 8-OHdG, IL-1β, IL-18, IL-6, and TNF-α. Information are offered as imply values ± SD. (n = 3). **p < 0.01 and ***p < 0.001
Given the promising therapeutic results of Wants demonstrated each in vitro and in vivo dry eye fashions, we carried out an in vivo security analysis to discover its translational potential (Fig. S15). The Wants have been administered twice every day (5 µL per dose) into the conjunctival sac for 7 consecutive days. Histological examination of ocular tissues (cornea, conjunctiva, iris, len, and retina) and main organs (coronary heart, liver, spleen, lung, and kidney) have been performed utilizing hematoxylin and eosin (H&E) staining. The outcomes demonstrated that Wants-treated mice exhibited regular mobile density and structure in ocular tissues, with clear stratification and regular thickness, akin to regular mice. Moreover, no histopathological abnormalities have been detected in any main organs. Collectively, these findings affirm the superb biocompatibility of Wants in vivo, supporting its potential as a protected and promising therapeutic technique for DED.