Picture-controlled co-delivery of Vp and Af through platelets achieved potentiated GBM PDT
Characterization of Vp@Plt, Af@Plt, Vp@Plt + Af@Plt, and (Vp + Af)@Plt
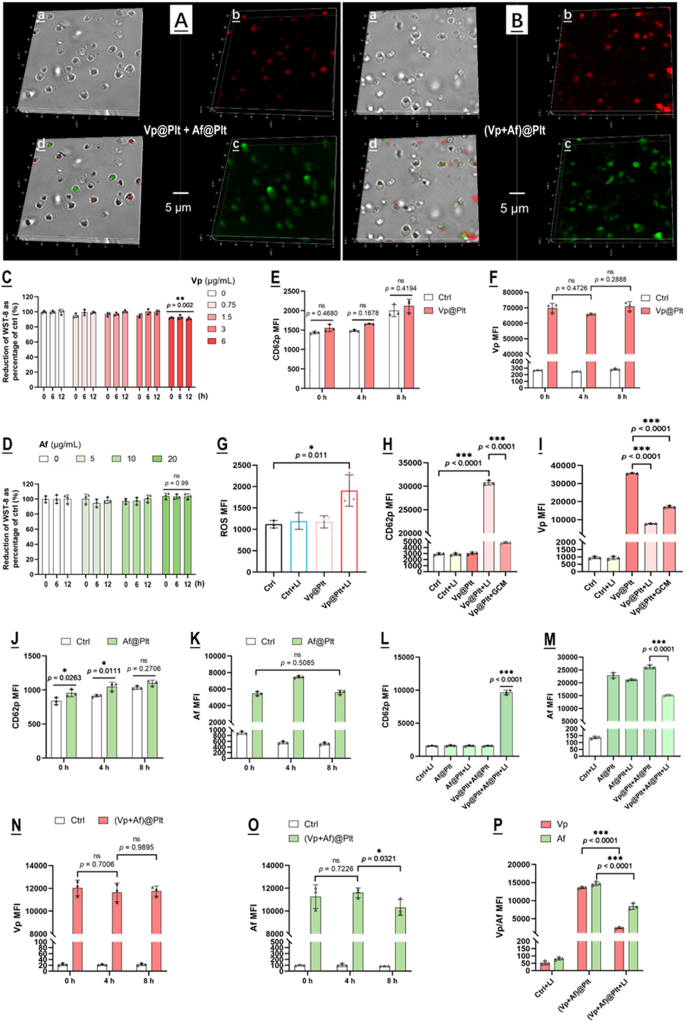
Vp@Plt and Af@Plt have been ready by incubating remoted mouse platelets (106/mL) individually with Vp (3 µg/mL in PBS) and Af (10 µM in PBS) for 30 min. (Vp + Af)@Plt have been ready by incubating mouse platelets with a combination of Vp (3 µg/mL) and Af (10 µM) in PBS for 30 min. The importing of Vp, Af, and Vp + Af have been confirmed by confocal microscopy (Fig. 1A, B). Vp@Plt solely exhibited Vp-derived fluorescence and Af@Plt solely exhibited Af-derived fluorescence whereas (Vp + Af)@Plt exhibited each Vp- and Af- derived fluorescence on the similar time (Fig. 1A, B). The non-toxic loading focus for Vp (3 µg/mL) and Af (10 µM) have been decided utilizing the CCK-8 take a look at to assay the viability of platelets after incubation with Vp or Af throughout a variety of concentrations (Fig. 1C, D). Beneath these situations, loading effectivity was 0.48 µg/106 platelets for Vp and 0.25 µg/106 platelets for Af. Vp@Plt have been fairly steady inside 8 h of preparation displaying no vital signal of activation and spontaneous launch as indicated by the expression of floor CD62p and mobile Vp fluorescence (Fig. 1E, F). Upon LI (690 nm, 0.5 W/cm2, 60 s), Vp@Plt exhibited a spike of ROS era with concurrent platelet activation and fast lack of Vp (Fig. 1G-I). Notably, the GBC-conditioned tradition medium (GCM) may additionally induce Vp@Plt activation and discharge of Vp, albeit to a a lot lesser extent (Fig. 1H, I). Alternatively, Af@Plt additionally maintained stability inside 8 h of preparation with out activation and spontaneous launch (Fig. 1J, Okay). Neither LI (690 nm) nor mixing with Vp@Plt had any outstanding impact on Af@Plt (Fig. 1L, M). Nevertheless, LI (690 nm) on the combination of Vp@Plt + Af@Plt effected marked platelet activation and fast lack of Af from the Af@Plt (Fig. 1L, M). Equally, (Vp + Af)@Plt remained steady displaying little signal of platelet activation and spontaneous offloading inside 8 h of preparation however exhibited a marked lack of Vp and Af upon LI (690 nm) (Fig. 1N-P).
Characterization of Vp@Plt, Af@Plt, Vp@Plt + Af@Plt, and (Vp + Af)@Plt. A & B: Confocal microscopy of Vp@Plt + Af@Plt, and (Vp + Af)@Plt. Vp fluorescence (purple) was noticed within the Cy5 channel with 650 nm excitation and emission at 670 nm. Af fluorescence (inexperienced) was noticed within the FITC channel with 488 nm excitation and emission at 520 nm. a: Brilliant area. b: Vp-derived fluorescence. c: Af-derived fluorescence. d: Merge of a, b, and c. C&D: Platelets incubated with Vp and Af at a variety of concentrations have been maintained in PBS and aliquots of the loaded platelets have been taken at completely different time factors for viability assay. E Spontaneous activation of Vp@Plt over time, indicated by floor CD62P expression. F: Spontaneous offloading from Vp@Plt over time, indicated by intracellular Vp content material. G: ROS era within the Vp@Plt upon LI (690 nm). H: Activation of Vp@Plt by LI (690 nm) and GBC-conditioned tradition medium (GCM). I: Offloading from Vp@Plt triggered by LI (690 nm) and GCM. J: Spontaneous activation of Af@Plt over time, indicated by floor CD62P expression. Okay: Spontaneous offloading from Af@Plt over time, indicated by intracellular Af content material. L: Activation of Vp@Plt + Af@Plt by LI (690 nm). M: LI (690 nm) triggered offloading of Af@Plt in combination with Vp@Plt however not Af@Plt solo. N&O: Spontaneous offloading from (Vp + Af)@Plt over time, indicated by intracellular Vp and Af content material. P: LI (690 nm) triggered offloading of (Vp + Af)@Plt. Values are means ± commonplace deviation (SD) (n = 3, *p < 0.05, **p < 0.01, ***p < 0.001). Consultant circulate cytometry contour plots for E-P are offered in Fig. S2
Picture-triggered, GBM-targeted supply mediated by Vp@Plt, Vp@Plt + Af@Plt, and (Vp + Af)@Plt
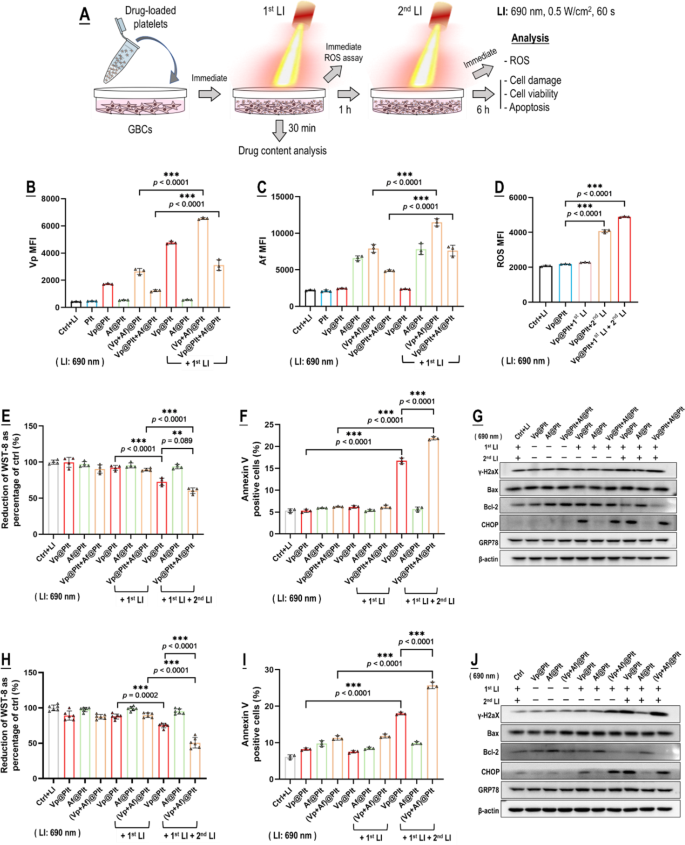
We subsequent demonstrated photo-controlled drug supply from Vp@Plt, Vp@Plt + Af@Plt, and (Vp + Af)@Plt to in vitro mouse GBCs (GL261) per an experimental protocol proven in Fig. 2A. Working focus was 0.48 µg/mL for Vp and 0.25 µg/mL for Af. The GBCs solely displayed a low content material of Vp, Af, Vp + Af, and Vp + Af following 1 h of therapy of Vp@Plt, Af@Plt, Vp@Plt + Af@Plt, and (Vp + Af)@Plt, respectively (Fig. 2B, C), possible resulting from a low degree of platelet activation and drug launch elicited by the GBCs (Fig. 1H, I). In distinction, LI (690 nm, 0.5 W/cm2, 60 s) on the outset of the therapy resulted in sharply elevated drug contents in all teams of GBCs however these handled with Af@Plt (Fig. 2B, C), indicating the incidence of LI-triggered supply of Vp and co-delivery of Vp & Af to the GBCs from the loaded platelets. The dearth of LI-triggered drug supply by Af@Plt alone (Fig. 2C) is affordable as LI (690 nm) doesn’t excite Af. Subsequent, the GBCs that had been handled with Vp@Plt exhibited pronounced ROS era upon a second LI 1 h after the primary LI (Fig. 2D), which led to vital cytotoxicity manifested as cell viability loss (WST-8), DNA harm (γH2AX), ER stress (CHOP, GRP78), and apoptosis (Annexin V, Bax & Bcl-2) (Fig. 2E-J). Considerably, photo-controlled co-delivery of Vp and Af by the use of Vp@Plt + Af@Plt (Fig. 2E-G) and (Vp + Af)@Plt (Fig. 2H-J) each led to markedly enhanced photo-cytotoxicity over Vp@Plt.
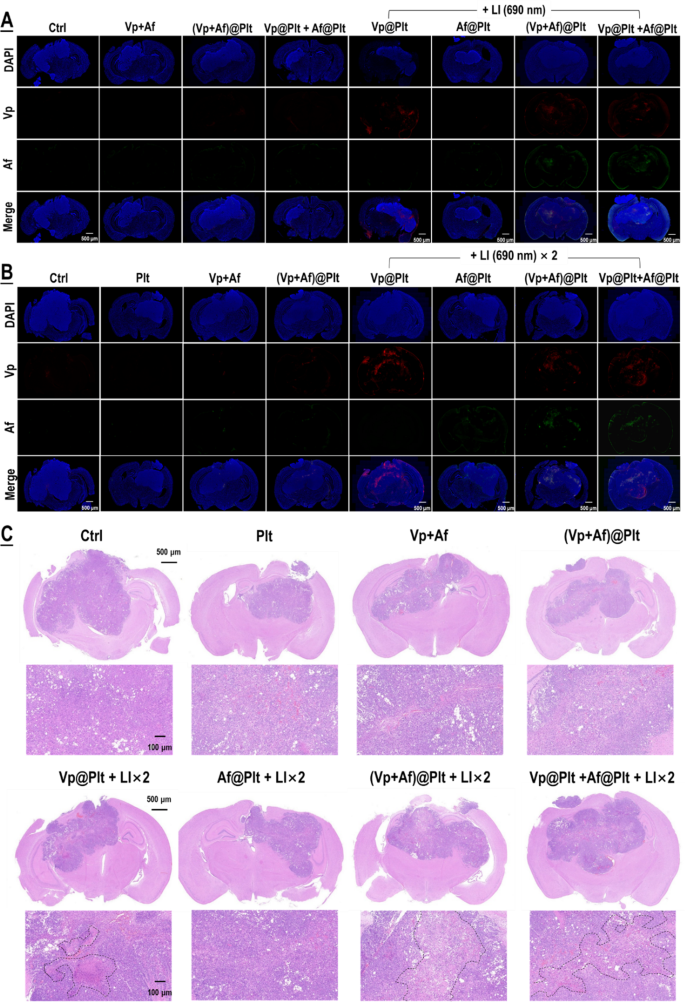
The in vitro outcomes have been then validated in mice bearing intra-cranial grafted GBMs per protocols proven in Fig. S4. The animals, in accordance with their assignments, have been every given an intravenous bolus injection of Vp@Plt, Af@Plt, (Vp + Af)@Plt, or Vp@Plt + Af@Plt, in 200 µL of PBS. The dosage was 24 µg/kg for Vp and 12.5 µg/kg for Af. After a drug-to-light interval of 1 h, two extra-cranial LI (690 nm, 0.5 W/cm2, 60 s) have been utilized to every animal on the tumor website at a 2-hr interval. The primary LI was meant to induce GBM-targeted, photo-controlled drug supply and the second LI was for activation of the Vp distributed within the tumor and thereby elicitation of photodynamic harm of the tumor. As proven in Fig. 3A, all animals however these injected with Af@Plt exhibited outstanding drug distribution within the GBMs 2 hrs after the primary LI. Notably, the mice injected with Vp@Plt solely displayed Vp fluorescence within the GBMs whereas the mice injected with both (Vp + Af)@Plt or Vp@Plt + Af@Plt displayed each Vp and Af fluorescence within the GBMs, indicating profitable co-delivery of the 2 brokers. The dearth of Af distribution within the GBMs within the Af@Plt-injected mice is consistent with the in vitro commentary proven in Fig. 2C, that are each ascribed to the truth that Af just isn’t excited by LI (690 nm). Additional alongside, elevated drug fluorescence indicating accumulation of delivered drug was noticed in all animals however these injected with Af@Plt 24 h after the second LI (Fig. 3B). In settlement with the drug supply profiles, the mice that had acquired (Vp + Af)@Plt or Vp@Plt + Af@Plt exhibited extra huge and extreme tissue necrosis within the GBMs than people who acquired Vp@Plt 24 h after the second LI, indicating that the co-delivered Af enhanced Vp-mediated photo-damage of the GBMs (Fig. 3C). In contrast, not one of the management animals that had acquired an intravenous injection of Vp + Af, or (Vp + Af)@Plt, or Vp@Plt + Af@Plt however with out LI displayed vital drug distribution, accumulation (Fig. 3A, B), and big tissue necrosis (Fig. 3C) within the GBMs. Nor did the mice that had acquired the Af@Plt injection plus LI current huge tissue necrosis within the GBMs they have been carrying (Fig. 3C).
Picture-triggered drug supply from Vp@Plt, Vp@Plt + Af@Plt, and (Vp + Af)@Plt to in vitro GBCs and subsequent photocytoxicity. A: Experimental protocol. Briefly, Vp@Plt, Af@Plt, Vp@Plt + Af@Plt, and (Vp + Af)@Plt have been individually combined with GL261 cells and the combination acquired two LI (690 nm, 0.5 W/cm2, 60 s) at an interval of 1 h. The GL261 cells have been taken out for evaluation at indicated time factors. B&C: Vp and Af contents within the GL261 cells after the primary LI, assayed by circulate cytometry. D: ROS era within the GL261 cells upon the first and 2nd LI. E–G: Picture-cytotoxicity of Vp@Plt + Af@Plt vs. Vp@Plt and Af@Plt indicated by WST-8 take a look at, annexin v staining, and expression of γH2AX, Bax, Bcl-2, CHOP, and GRP78. H–J: Picture-cytotoxicity of (Vp + Af)@Plt vs. Vp@Plt and Af@Plt indicated by WST-8 take a look at, annexin v staining, and expression of γH2AX, Bax, Bcl-2, CHOP, and GRP78. Values are means ± commonplace deviation (SD) (n = 3, *p < 0.05, **p < 0.01, ***p < 0.001). Consultant circulate cytometry contour plots for E-P are offered in Fig. S3. Quantitative evaluation of Western blot knowledge in G & J is proven in Fig. S12 & Fig. S13 within the complement data
Picture-triggered, GBM-targeted drug supply by Vp@Plt, Vp@Plt + Af@Plt, and (Vp + Af)@Plt and subsequent phototoxicity to the GBM. Experimental protocol is proven in Fig. S3. Briefly, intracranial GBM-bearing mice have been intravenously injected with Vp@Plt, Af@Plt, Vp@Plt + Af@Plt, or (Vp + Af)@Plt and, 1 h later, acquired 2 LI (690 nm, 0.5 W/cm2, 60 s) on the tumor website at a 2-hr interval. A part of the animals have been sacrificed 2 hr after the first LI and the remaining sacrificed 24 h after the 2nd LI. The brains have been taken for fluorescent staining, H&E staining and microscopy. A: Vp and Af fluorescence within the GBMs 2 hr after the first LI. B: Vp and Af fluorescence within the GBMs 24 h after the 2nd LI. C: H&E staining of the mind and GBM tissues harvested 2 hr after the first LI and 24 h after the 2nd LI. Areas circled within the dotted strains are tissues with extreme necrosis. Consultant increased magnification confocal fluorescent microscopic photographs of are offered in Fig. S5 & S6
Vp@Plt + Af@Plt and (Vp + Af)@Plt achieved stronger GBM-targeted PDT than Vp@Plt
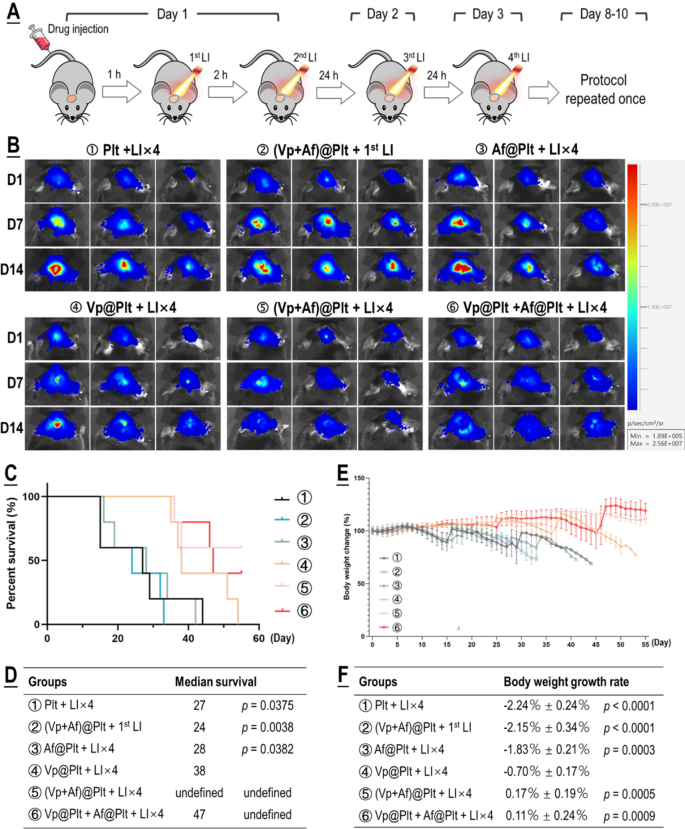
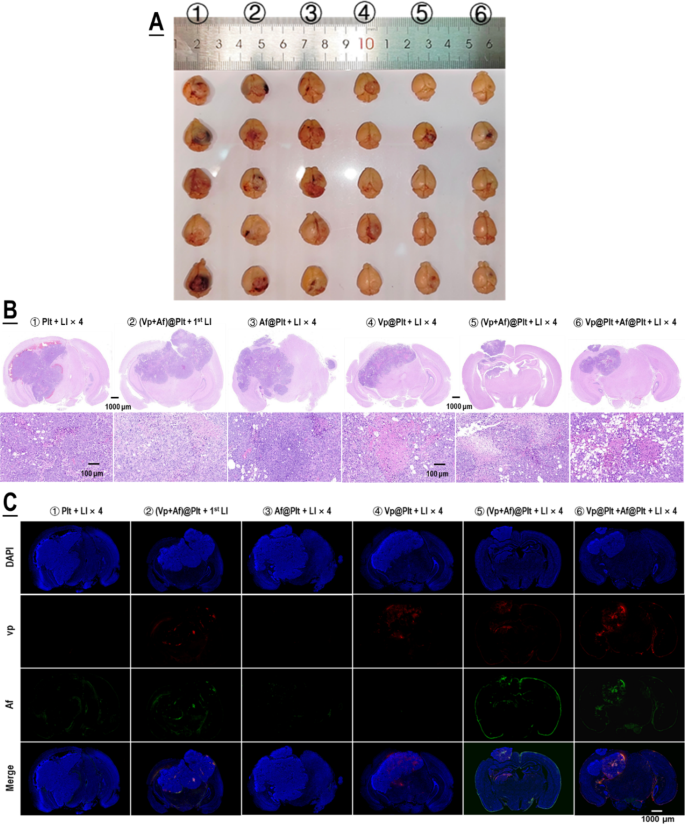
For analysis of therapeutic efficacy, mice bearing intra-cranial grafted GBMs have been handled per a protocol illustrated in Fig. 4A. Briefly, the GBM-bearing mice which have been divided into six teams, in accordance with their assignments, have been every given an intravenous bolus injection of ②Plt, ②(Vp + Af)@Plt, ③Af@Plt, ④Vp@Plt, ⑤(Vp + Af)@Plt, or ⑥Vp@Plt + Af@Plt, in 200 µL of PBS. The dosage was 24 µg/kg for Vp and 12.5 µg/kg b.w. for Af. After a drug-to-light interval of 1 h, the animals in teams ②, ③, ④, ⑤, and ⑥ every acquired 2 LI (690 nm, 0.5 W/cm2, 60 s) on the tumor website at a 2-hr interval. Two additional LI have been carried out, one 24 h and the opposite 48 h after the second LI. The animals in group ② solely acquired the primary LI 1 h after drug injection. The primary LI was meant for GBM-targeted, photo-triggered drug supply and all the opposite LI have been for activation of the Vp distributed within the tumor and thereby elicitation of photodynamic harm of the tumor. The therapy protocol was carried out twice at an interval of 5 days. As proven in Fig. 4B-F, Vp@Plt- mediated PDT (Vp@Plt + LI×4, group ④) confirmed outstanding anti-GBM efficacy with such manifestations as markedly slowed tumor progress, vital extension of host survival, and far alleviated physique weight reduction. Extra importantly, each (Vp + Af)@Plt (group ⑤) and Vp@Plt + Af@Plt (group ⑥) achieved tangibly increased therapeutic efficacy than Vp@Plt (group ④). Notably, 3 out of the 5 animals in group ⑤ that acquired (Vp + Af)@Plt + LI×4 and a couple of out of the 5 animals in group ⑥ that acquired Vp@Plt + Af@Plt + LI×4 have been nonetheless alive on the finish of the experiment by which era all in any other case handled animals had already been misplaced. In distinction, neither (Vp + Af)@Plt + 1st LI (group ②) nor Af@Plt + LI×4 (group ④) exhibited considerable therapeutic efficacy. Moreover, gross examination of resected brains confirmed smaller tumor growths and considerably higher preserved mind morphology in teams ④, ⑤ and ⑥ (Fig. 5A). H&E staining additionally revealed huge tissue necrosis within the tumor grafts in teams ④, ⑤ and ⑥, and there was no obvious mind tissue harm (Fig. 5B). Remarkably, tumor grafts in teams ②, ④, ⑤ and ⑥, on the time of host sacrifice which was 25 days after the final time of drug administration, nonetheless displayed considerable drug retention as indicated by the drug-derived fluorescence (Fig. 5C). These observations are compelling proof that (1) photo-controlled supply of Vp through Vp@Plt achieved efficacious anti-GBM PDT, (2) photo-controlled co-delivery of Vp and Af, both by (Vp + Af)@Plt or Vp@Plt + Af@Plt achieved stronger anti-GBM PDT than Vp@Plt.
Anti-GBM efficacy of PDT mediated by photo-activated Vp@Plt, Vp@Plt + Af@Plt, and (Vp + Af)@Plt. A: Experimental protocol. Briefly, 5 teams of GBM-bearing animals have been intravenously injected with platelets (Plt), Af@Plt, Vp@Plt, (Vp + Af)@Plt, and Vp@Plt + Af@Plt, respectively (①③④⑤⑥). The animals have been then subjected to a succession of 4 LI (0.5 W/cm2, 60 s) at intervals on the tumor website. The therapy protocol took 3 days to finish and was repeated as soon as. As management, one other group of animals have been injected with (Vp + Af)@Plt and solely acquired the first LI (②). B: Fluorescent photographs of mind tumors in vivo taken on day 1, 7, and 14 into remedy. GBM-derived luminescence on day 1 and the rise in GBM-derived luminescence as much as day 14 was quantified and confirmed in Fig. S7. C & D: Survival evaluation of handled animals. E & F: Physique weight of handled animals monitored over the remedy length. Physique weight progress charges have been calculated by regression evaluation. Values have been means ± SD (n = 5)
Each Af and Vp may potentiate GBM PDT by blocking PDT-induced interplay of HIF-1α and YAP in GBCs
Each Af and Vp potentiated photodynamic toxicity in GBCs
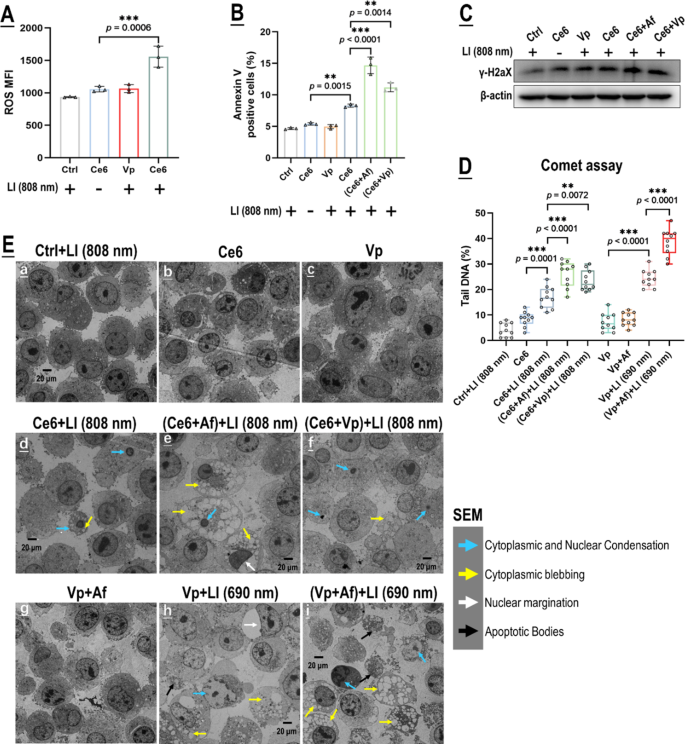
We subsequent got down to display our authentic mechanistic speculation that Af may potentiate the photo-cytotoxicity of Vp-mediated PDA by blocking the induced HIF-1α. Vp, nevertheless, has pharmacological actions (e.g. inhibition of YAP) [38, 39] that might possible bear on its photo-cytotoxicity and the exercise of Af. Contemplating this chance, we adopted one other photosensitizer, chlorin e6 (Ce6) plus 808 nm of excitation mild (Ce6 + LI(808)) in parallel with Vp + LI(690 nm) within the mechanistic research. We had beforehand demonstrated that Ce6 may be excited by LI(808 nm) which doesn’t excite Vp [37]. Herein, as proven in Fig. 6A, Ce6 generated ROS within the GBCs beneath LI(808 nm) however Vp didn’t. Af as anticipated potentiated Ce6’s photo-toxicity to the GBCs beneath LI(808 nm), as was indicated by a marked improve in DNA harm (γH2X & comet assay) and apoptosis (floor annexin v) (Fig. 6B-D). Surprisingly, Vp additionally potentiated the photo-cytotoxicity of Ce6 beneath LI(808 nm) (Fig. 6B-D). TEM offered additional proof that each Af and Vp enhanced GBC apoptosis induced by Ce6 + LI(808 nm) (Fig. 6E). These findings recommend that (1) Vp has non-photosensitizing results that may potentiate the photo-toxicity of itself and different photosensitizers; and (2) the potentiative motion of Af won’t be restricted to Vp-mediated PDA but in addition apply to different photosensitizers.
Each Af and Vp potentiated the photo-toxicity of Ce6 beneath LI (808 nm). A: Ce6 however not Vp generated ROS beneath LI (808 nm) in in vitro GL261 cells. Impact of Vp, Ce6, Ce6 + Af, and Ce6 + Vp beneath LI (808 nm) on cell floor annexin v expression (B), γH2AX (C), DNA harm indicated by the comet assay (D), and cell morphology noticed through SEM (E). Consultant circulate cytometry contour plots for A & B are proven in Fig. S9 A & B. Comet assay photographs for D are proven in Fig. S9C
PDA induced HIF-1α, which each could possibly be suppressed by Af and Vp
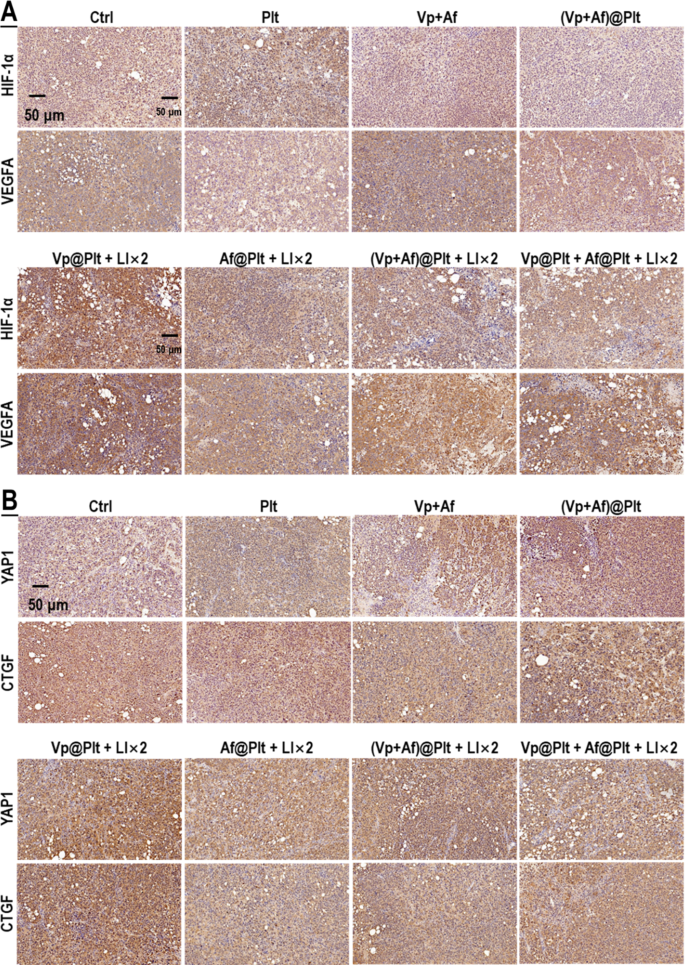
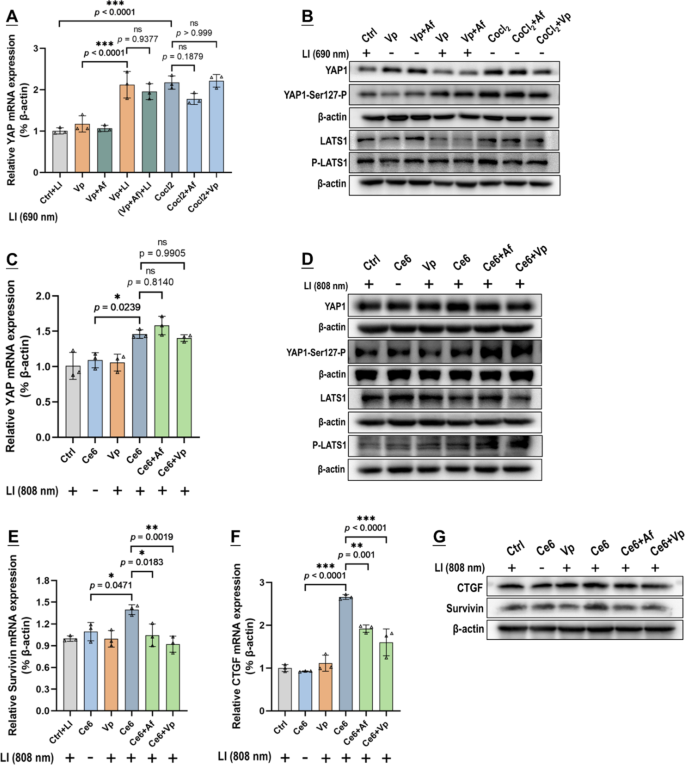
As talked about above, our preliminary speculation was that Af may potentiate the photo-cytotoxicity of Vp + LI(690 nm) by repressing the induced HIF-1α. Thus, to start with, we examined the impact of Af on the gene transcription (mRNA degree), protein content material, degradation exercise (indicated by the degrees of PHD2 and P-VHL) of HIF-1α, and its downstream targets in in vitro GBCs following publicity to Vp + LI (690 nm). CoCl2, a broadly used hypoxia-mimicking agent that will increase HIF-1α by inhibiting the PHD [40], was used for management. As proven in Fig. 7A, HIF-1α mRNA degree confirmed little change following publicity to Vp + LI(690 nm). Paradoxically, there was an apparent improve of HIF-1α protein and its downstream targets (VEGFA, GLUT1, & CA9) (Fig. 7B-E). This obvious inconsistency may solely be attributed to decreased HIF-1α degradation as was certainly indicated by a discount in PHD2 and P-VHL, the principal enzymes chargeable for HIF-1α degradation (Fig. 7F). Af, as anticipated, alleviated the upregulation of HIF-1α and its downstream targets induced by Vp + LI (690 nm) (Fig. 7E). CoCl2 additionally upregulated HIF-1α and its downstream targets (Fig. 7E) with persistently decreased PHD2 and P-VHL (Fig. 7F) and these results weren’t solely alleviated by Af however, apparently, additionally by Vp (Fig. 7E, F). This commentary means that Vp could have non-photosensitizing results on the PDA-induced HIF-1α and this assumption was substantiated by means of Ce6 + LI(808 nm). As proven in Fig. 7G-Okay, Ce6 + LI(808 nm), like Vp + LI(690 nm), had little impact on HIF-1α mRNA degree however resulted in a outstanding improve in HIF-1α protein and its downstream targets (VEGFA & CA9), in addition to decreased PHD2 and P-VHL indicative of diminished HIF-1α degradation. Considerably, each Af and Vp markedly suppressed the upregulation of HIF-1α protein and its downstream targets with little bearing on HIF-1α mRNA (Fig. 7G-J), and this impact was accompanied by a rise in P-VHL indicating recovered HIF-1α degradation (Fig. 7Okay). The in vitro outcomes have been nicely mirrored within the in vivo orthotopic GBMs uncovered to Vp@Plt + LI (690 nm), which exhibited enhanced expression of HIF-1α and VEGFA, which was alleviated by Af delivered both within the type of (Vp + Af)@Plt or Vp@Plt + Af@Plt (Fig. 8A). To recap briefly, PDA induced HIF-1α by decreasing its degradation slightly than upregulating its gene transcription, and this impact could possibly be antagonized by each Af and Vp resulting in elevated HIF-1α degradation.
Each Vp and Af suppressed HIF-1α induction brought on by Vp beneath LI (690 nm) or Ce6 beneath LI(808 nm). Impact of Vp and Af on mRNA ranges of HIF-1α (A), VEGFA (B), GLUT1 (C), and CA9 (D), their protein ranges (E), and ranges of HIF-1α degradation enzymes PHD2 and P-VHL(F) in G261 cells uncovered to PDA mediated by Vp beneath LI(690 nm). CoCl2 was used as a management therapy in A–F. Impact of Vp and Af on mRNA ranges of HIF-1α (G), VEGFA (H), and CA9 (I), their protein ranges (J), and ranges of HIF-1α degradation enzymes PHD2 and P-VHL (Okay) in G261 cells uncovered to PDA mediated by Ce6 beneath LI (808 nm). Values have been means ± SD (n = 3, *p < 0.05, **p < 0.01, ***p < 0.001). Quantitative evaluation of Western blot knowledge in F is proven in Fig. S14 within the complement data
Af delivered within the type of (Vp + Af)@Plt and Af@Plt + Vp@Plt each suppressed the induction of HIF-1α and YAP brought on by Vp@Plt beneath LI (690 nm) in intracranial GBM tissues. Experimental protocol is proven in Fig. S4. Briefly, intracranial GBM-bearing mice have been intravenously injected with Vp@Plt, Af@Plt, Vp@Plt + Af@Plt, or (Vp + Af)@Plt and, 1 h later, acquired 2 LI (690 nm, 0.5 W/cm2, 60 s) on the tumor website at a 2-hr interval. The animals have been sacrificed 24 h after the 2nd LI. The brains have been taken for immunohistochemical (IHC) staining of HIF-1α, VEGFA (A), YAP, and CTGF (B)
PDA induced YAP, which may each be suppressed by Vp and Af
In mild of the above observations and the truth that Af is a acknowledged HIF-1α inhibitor and Vp is a widely known YAP inhibitor [25, 26], we suspected that PDA may induce an interaction of HIF-1α and YAP within the GBCs. To discover this notion, we surveyed the gene transcription (mRNA degree), protein content material, and degradation exercise of YAP in in vitro GBCs uncovered to PDA. Beneath Vp + LI (690 nm), there was an upsurge in YAP mRNA however, paradoxically, no improve in YAP protein (Fig. 9A & B). This inconsistency may solely be defined by elevated YAP degradation which was clearly indicated by a marked improve in YAP-Ser127-P and P-LATS1 (Fig. 9B). (Notice: P-LATS1 is the activated shaped of LATS1, which triggers YAP phosphorylation at Ser127 (YAP-Ser127-P), leading to cytoplasmic retention and degradation [41]). Vp, an agent identified to advertise YAP degradation [38, 39], was strongly indicated to be the actor underlying the decreased YAP protein by one other two strains of proof. First, Vp alleviated CoCl2-induced YAP protein degree with out affecting YAP mRNA (Fig. 9A & B). Second, Ce6 + LI(808 nm) additionally induced YAP and its downstream targets (Survivin & CTGF) as nicely and Vp alleviated the induced protein degree of YAP and its downstream targets with out affecting YAP mRNA (Fig. 9C-G). Considerably, Af additionally repressed the induced YAP protein degree and its downstream targets with out affecting YAP mRNA (Fig. 9C-G), and each Vp and Af resulted in heightened YAP degradation as was indicated by a marked improve in YAP-Ser127-P and P-LATS1 (Fig. 9D). The orthotopic GBMs displayed enhanced YAP and CTGF expression following publicity of Vp@Plt + LI (690 nm) (Fig. 8B), which seemed to be inconsistent with the noticed YAP lower in in vitro GBCs (Fig. 9B). This inconsistency, as is mentioned later, is attributed to the aggravated hypoxia within the tumor tissue because of PDT. However, the improved YAP and CTGF expression was alleviated by Af delivered both within the type of (Vp + Af)@Plt or Vp@Plt + Af@Plt (Fig. 8B), which is consistent with the in vitro findings. To recap briefly, PDA induced YAP through upregulating its gene expression, which impact could possibly be antagonized each by Vp and Af resulting in elevated YAP degradation.
A synthesis of the findings in subsections 2.2 and 2.3 recommend that PDA may induce an interaction of HIF-1α and YAP, which begins with heightened YAP gene transcription and results in diminished degradation of each elements; each Af and Vp may block this interplay subsequently rising the degradation of HIF-1α and YAP. There was research suggesting that HIF-1α can bodily work together with YAP in most cancers and non-cancer cells beneath hypoxic stress, selling the capabilities of each elements [34, 42, 43]. We puzzled PDA may induce an identical interplay in GBCs and located related proof which is offered beneath.
Each Vp and Af suppressed YAP induction brought on by Vp beneath LI (690 nm) or Ce6 beneath LI(808 nm). Impact of Vp and Af on YAP mRNA degree (A), protein ranges of YAP, YAP-Ser127-P, LATS1, and P-LATS1 (B) in G261 cells uncovered to PDA mediated by Vp beneath LI (690 nm). CoCl2 was used as a management therapy in A & B. Impact of Vp and Af on YAP mRNA degree (C), protein ranges of YAP, YAP-Ser127-P, LATS1, and P-LATS1 (D), mRNA ranges of Survivin (E) and CTGF (F), and their protein ranges (G) in G261 cells uncovered to PDA mediated by Ce6 beneath LI(808 nm). (n = 3, *p < 0.05, **p < 0.01, ***p < 0.001). Quantitative evaluation of Western blot knowledge in B & D is proven in Fig. S15 within the complement data
PDA elicited an interaction of HIF-1α and YAP which may each be blocked by Af and Vp
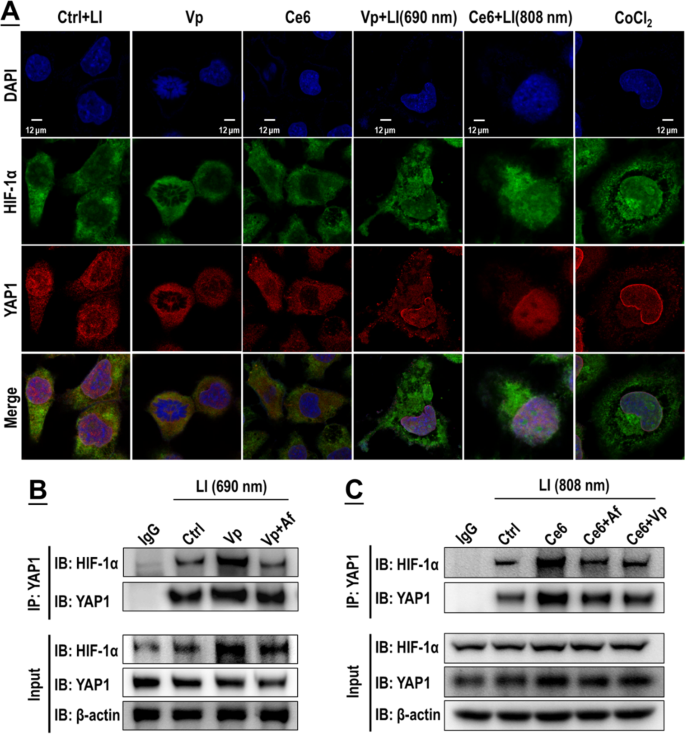
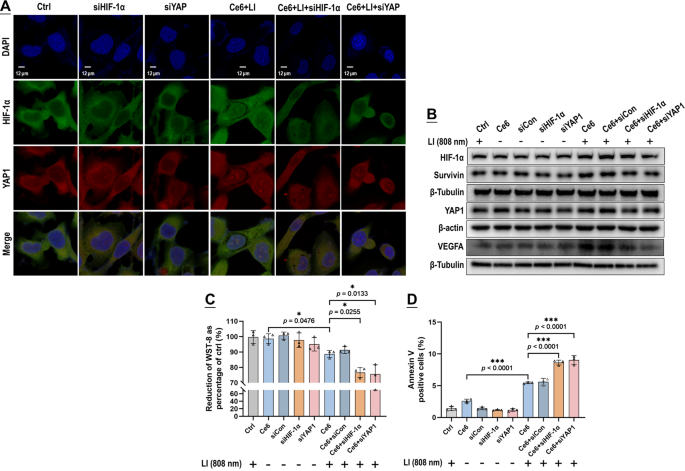
The GBCs exhibited a conspicuous nuclear co-localization of HIF-1α and YAP following publicity of Vp + LI (690 nm), or Ce6 + LI (808 nm), or CoCL2 (Fig. 10A). Co-IP assay additionally recognized elevated binding of HIF-1α and YAP each beneath Vp + LI (690 nm) and Ce6 + LI (808 nm), which could possibly be each suppressed by Vp and Af (Fig. 10B, C). Notably, the Co-IP assay additionally demonstrated the adjustments in HIF-1α and YAP protein ranges on the similar time following publicity to PDA (Fig. 10B, C). Each Vp + LI (690 nm) and Ce6 + LI (808 nm) triggered a rise of HIF-1α which could possibly be alleviated by AF. However YAP behaved in a different way. Beneath Vp + LI (690 nm), the YAP degree declined, which was ascribed to Vp-mediated degradation and compounded by the addition of Af (Fig. 10B). Beneath Ce6 + LI (808 nm), the YAP degree elevated, which could possibly be each suppressed by Af and Vp (Fig. 10C). General, these observations are per these proven in Figs. 7 and 9. Silencing of HIF-1α and YAP by siRNA produced an additional line of proof of an HIF-1α-YAP interaction induced by PDA. Knock-down of HIF-1α and YAP each alleviated the nuclear co-localization of HIF-1α and YAP elicited by Ce6 + LI (808 nm) (Fig. 11A) and suppressed the induction of HIF-1α, YAP, and their downstream targets (Fig. 11B). Additionally, WST-8 assay (Fig. 11C) and cell floor annexin v assay (Fig. 11D) indicated that knock-down of HIF-1α and YAP each potentiated the photo-cytotoxicity of Ce6 + LI (808 nm) very similar to Af and Vp did (Fig. 6).
To recap, PDA elicited an interplay of HIF-1α and YAP which boosted their nuclear distribution and thereby elevated the expression of their downstream targets. Af or Vp may block this interplay impacting each HIF-1α and YAP and potentiate the PDA-induced toxicity within the GBCs. In mild thereof, we posited that the HIF-1α-YAP interplay is perhaps a part of the mobile response to PDA-inflicted cell harm, which is mobilized for harm restore and sustaining cell survival. Proof is as follows.
Vp beneath LI (690 nm) and Ce6 beneath LI (808 nm) each stimulated an interaction of HIF-1α and YAP, which could possibly be blocked by Vp or Af. A: Enhanced fluorescent co-localization of HIF-1α and YAP within the nucleus. CoCl2 was used as a management therapy. B&C: Co-immunoprecipitation (CO-IP) evaluation indicated elevated binding of HIF-1α and YAP, which impact could possibly be each suppressed by Vp and Af
Knock-down of HIF-1α and YAP each prevented the interaction of HIF-1α and YAP stimulated by Ce6 beneath LI (808 nm) and potentiated the photo-cytotoxicity. Knock-down of HIF-1α and YAP each alleviated fluorescent co-localization of HIF-1α and YAP within the nucleus (A), decreased the expression of HIF-1α, VEGFA, YAP, and Survivin (B), and led to enhanced photo-cytotoxicity indicated by WST-8 assay (C) and floor annexin v staining (D). Values have been means ± SD (n = 3, *p < 0.05, ***p < 0.001). Consultant circulate cytometry contour plots for D are proven in Fig. S10
PDA-elicited HIF-1α-YAP interplay resulted from DNA harm and led to upregulated DNA restore exercise
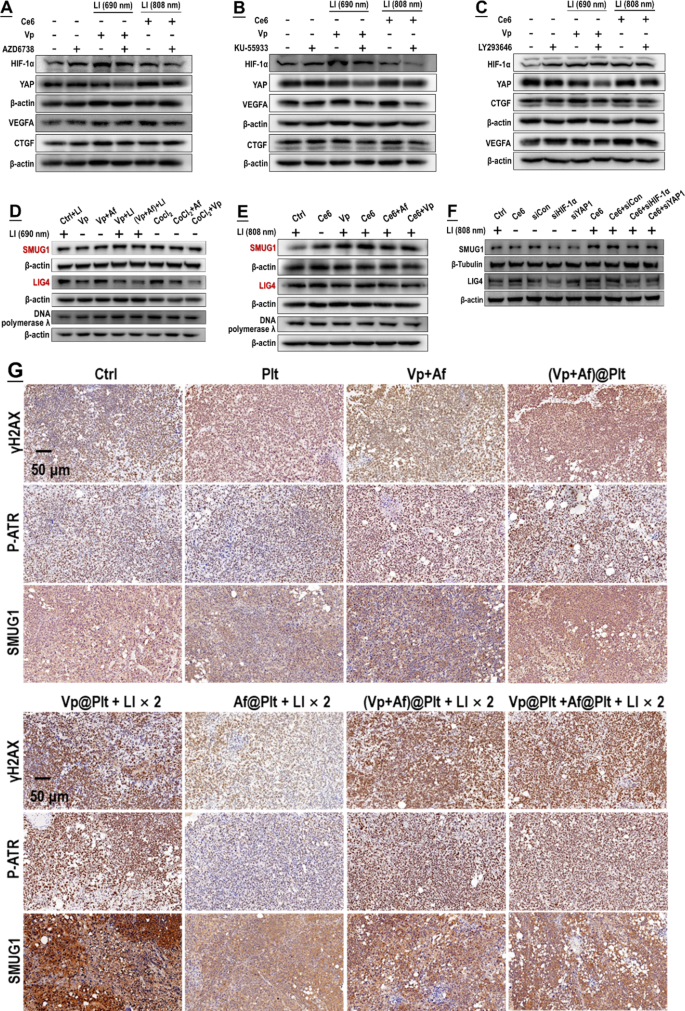
As pronounced DNA harm was recognized following publicity to PDA (Fig. 2), we blocked the DNA harm response in PDA-exposed GBCs using pharmacologic inhibitors of the ATR, ATM, and DNA-PK, that are main DNA harm sensors [44, 45]. As proven in Fig. 12A-C, inhibition of every of the three sensors may suppress the upregulation of HIF-1α and YAP and their downstream targets both induced by Vp + LI (690 nm) or Ce6 + LI (808 nm). Alternatively, SMUG1 and DNA ligase IV (LIG4) are key enzymes concerned in nuclear DNA restore [46, 47]. Each Af and Vp markedly suppressed the induction of the 2 enzymes both brought on by Vp + LI (690 nm), or Ce6 + LI (808 nm), or CoCl2 (Fig. 12D-E). Persistently, knock-down of HIF-1α and YAP each suppressed the induction of SMUG1 and LIG4 brought on by Ce6 + LI (808 nm) (Fig. 12F). In brief, the HIF-1α-YAP interplay was elicited for mobilization of DNA restore which could possibly be each blunted by Af and Vp.
Induction of HIF-1α and YAP by PDA was a downstream occasion to DNA harm and led to upregulation of DNA harm restore exercise. A–C: Pharmacological blocking of DNA harm sensors (i.e. ATR, ATM, and DNA-PK) alleviated induction of HIF-1α and YAP each brought on by Vp beneath LI (690 nm) and Ce6 beneath LI(808 nm). AZD6738, KU-55,933, and LY293646 are inhibitors of the ATR kinase, ATM kinase, and DNA-PK, respectively. D&E: Each Af and Vp blocked induction of DNA restore enzymes (i.e. SMUG1 and LIG4) brought on by Vp beneath LI(690 nm) or Ce6 beneath LI(808 nm). F: Knock-down of HIF-1α and YAP each prevented the induction of SMUG1 and LIG4 brought on by Ce6 beneath LI(808 nm). G: Af delivered within the type of (Vp + Af)@Plt and Af@Plt + Vp@Plt each suppressed the induction of SMUG1 and LIG4 brought on by Vp@Plt beneath LI(690 nm) and potentiated the DNA harm (indicated by γH2AX and P-ATR expression) in intracranial GBM tissues. Experimental protocol is proven in Fig. S4. Briefly, intracranial GBM-bearing mice have been intravenously injected with Vp@Plt, Af@Plt, Vp@Plt + Af@Plt, or (Vp + Af)@Plt and, 2 h later, acquired 2 LI (690 nm, 0.5 W/cm2, 60 s) on the tumor website at a 2-hr interval. The animals have been sacrificed 24 h after the 2nd LI. The brains have been taken for IHC staining of γH2AX, P-ATR, SMUG1, and LIG4